| Clin Mol Hepatol > Volume 18(3); 2012 > Article |
See the commentary-article "Impact of ribavirin dose reduction during treatment in chronic hepatitis C genotype 1 patients" on page 268.
ABSTRACT
Background/Aims
When combined with pegylated interferon alpha-2b (Peg-IFN α-2b) for the treatment of genotype 1 chronic hepatitis C (CHC) in Korea, the current guideline for the initial ribavirin (RBV) dose is based on body weight. However, since the mean body weight is lower for Korean patients than for patients in Western countries, current guidelines might result in Korean patients being overdosed with RBV.
Methods
We retrospectively reviewed the medical records of patients with genotype 1 CHC who were treated with Peg-IFN α-2b and RBV combination therapy. We divided the patients into groups A (≥15 mg/kg/day, n=23) and B (<15 mg/kg/day, n=26), given that the standard dose is 15 mg/kg/day. The clinical course in terms of the virologic response, adverse events, and dose modification rate was compared between the two groups after therapy completion.
Results
The early response rates (92.0% vs. 83.3%, P=0.634) and sustained virologic response rates (82.6% vs. 73.1%, P=0.506) did not differ significantly between the two groups. During the treatment period, the RBV dose reduction rate was significantly higher in group A than in group B (60.9% vs. 23.1%, P=0.01).
Conclusions
RBV dose reduction is performed frequently when patients are treated according to the current Korean guidelines. Given that lowering the RBV dose did not appear to decrease the virologic response during therapy, reducing RBV doses below the current Korean guideline may be effective for treatment, especially in low-weight patients.
The hepatitis C virus (HCV) is a major public health problem and a leading cause of chronic liver disease. An estimated 180 million people are infected worldwide.1 The treatment for chronic hepatitis C (CHC) has evolved from the use of interferon alfa monotherapy to a combination of weekly polyethylene glycole interferon (Peg-IFN) plus weight-based ribavirin (RBV), as standard of care.2,3 Independent of viral genotype, it has been suggested that Asians are more likely than Caucasians to achieve a sustained virologic response (SVR) to the treatment of CHC with either interferon or pegylated interferon alpha in combination with RBV.4,5 In accordance with these observations, Korean patients with CHC have a high likelihood of responding to combination antiviral therapy with interferon and RBV.6,7 When patients were treated with Peg-IFN α-2b (1.5 µg/kg/week) plus RBV (1,000-1,200 mg/day for 48 weeks), SVR was achieved in 60-70% of patients with genotype 1.8 In Korea, the mean body weight of patients is relatively lower than in western countries. In low-weight patients, higher doses of RBV may result in several complications and, consequently, the patient's compliance may go worse or dose reduction may be required frequently. If the RBV dose is lowered the virologic response may drop, but it was reported that when Peg-IFN was administered sufficiently the virologic response did not drop even if the RBV dose was somewhat lower than the standard.9 Considering lower body weight in Korean patients, it is possible that RBV may be initially over-dosed using current weight-based Korean guidelines. Thus, this study purposed to analyze RBV dose administered according to weight and to examine the frequency of complications, virologic response, dose reduction rate, etc. in patients administered with RBV at a high dose per kg.
We retrospectively reviewed the medical records of patients with genotype 1 CHC, who were treated with the combination therapy of Peg-IFN α-2b (subcutaneous injection of 1.5 µg per kg body weight per week) and RBV based on body weight (1,000 mg for patients weighing <75 kg or 1,200 mg for patients weighing ≥75 kg), from October 2004 to February 2010, at Soon Chun Hyang University, Bucheon Hospital and Cheonan Hospital. The inclusion criteria for this study included men and women aged over 18 years, with serologically proven and treatment-naive CHC. In addition, the patients were required to have had detectable HCV RNA and compensated liver disease. Patients with liver cirrhosis and other forms of liver disease or other severe underlying diseases, i.e. cardiovascular disease and neoplasm, such as hepatocellular carcinoma, were excluded from our study. The initial RBV dose was defined as the cumulative RBV dose taken during the first 4 weeks converted to dosage per body weight per day. Based on their initial RBV dose, all patients were divided into two groups: Group A (≥15 mg/kg/day) and Group B (<15 mg/kg/day). Medical records of the study patients were reviewed to obtain baseline demographic features and clinical course after treatment with combination therapy. The primary objective of our study was to compare SVR, early virologic response (EVR) and dose reduction rates between the two groups following Peg-IFN α-2b plus RBV administered over 48 weeks.
HCV-RNA was determined in each participating center using commercially available polymerase chain reaction assays (Amplicor HCV or Amplicor HCV Monitor, Roche Diagnostics, Basel, Switzerland). Only patients with baseline and 12- or 24-week HCV-RNA determinations were included in the statistical analyses. The EVR was defined as ≥2 log reduction or complete absence of serum HCV RNA at week 12 of therapy compared with the baseline level. The SVR was defined as undetectable HCV-RNA (<50 IU per mL) 24 weeks after discontinuation of therapy.
Per protocol, the RBV dose had to be reduced by 200 mg daily in patients who experienced a decrease in hemoglobin level of more than 2 g/dL during treatment, and by 400 mg daily if hemoglobin levels decreased to less than 10 g/dL. RBV was discontinued if a patient without significant cardiovascular disease experienced a decrease in hemoglobin to a level below 8.5 g/dL or if a patient with stable cardiovascular disease maintained a hemoglobin value <12 g/dL, despite of being on a reduced dose of 600 mg for 4 weeks. Hematopoietic growth factors, such as erythropoietin, have been shown to improve the quality of life, but not SVR rates. In this study, we reduced the RBV dose instead of treatment with erythropoietin for patients with anemia. Further dose reduction for other adverse events was based on the severity of the side-effects, including myalgia and flu-like symptoms. However, the use of erythropoietin was not allowed. We decreased Peg-IFN α-2b dose for patients who developed neutropenia. Given that the dose of Peg-IFN α-2b is more vital in achieving SVR than the RBV dose, we only included in this study patients who had been treated for 48 weeks or more, to reduce the impact of the Peg-IFN α-2b dose.
Data were analyzed with SPSS software for Windows version 16.0 (Chicago, IL). Continuous variables are expressed as mean±standard deviation and categorical variables as absolute and relative frequencies. Mean differences were tested with the Student t-test and the Chi-square test for dichotomous and categorical variables. The result of analysis was considered statistically significant for a P-value <0.05.
A total of 49 patients (27 males and 22 females), who were treated for 48 weeks or more, were enrolled in this study. Based on the initial RBV dose, these patients were divided into two groups: Group A (≥15 mg/kg/day, n=23) and Group B (<15 mg/kg/day, n=26). Table 1 shows the baseline characteristics of the two groups. The mean body weight (57.2 vs. 70.5 kg, P=0.001), the mean body mass index (21.8 vs. 25.1 kg/m2, P=0.001) and the percentage of males (30.4 vs. 76.9%, P=0.020) in Group B was significantly higher than in Group A. The initial RBV dose (17.2 vs. 12.7 mg/kg/day, P=0.001) of patients in Group A was significantly higher than that in Group B. No significant difference was found in the mean age, alanine aminotransferase (ALT) and serum HCV RNA titers between the two groups. The mean duration (49.6 vs. 48.5 weeks, P=0.416) and dose reduction rate (30.4 vs. 23.1%, P=0.560) of PEG-IFN α-2b treatment showed no significant difference between the two groups. Among the patients who were treated with dose-reduced PEG-IFN α-2b treatment, one patient of Group A and two patients of Group B were treated with 40% dose reduced PEG-IFN α-2b and the others were treated with 20% dose reduced PEG-IFN α-2b.
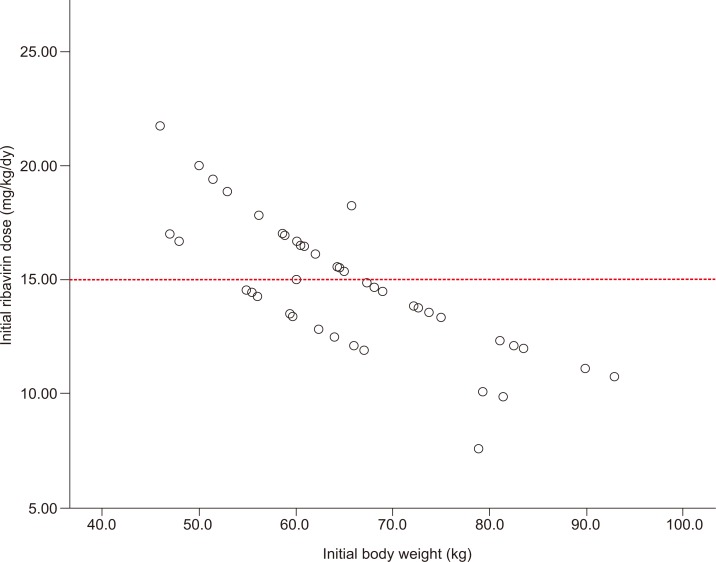
We also analyzed the relationship between the body weight of patients and the initial RBV dose. Most of the patients enrolled in our study (81.6%) weighed less than 70 kg, and half of these patients weighed 60 kg or less. The initial RBV dose (15.69 vs. 11.02 mg/kg/day, P=0.001) of patients whose body weight was under 75 kg was significantly higher than in patients whose body weight was over 75 kg. A total of 23 patients were treated with an initial RBV dose of 15 mg/kg/day or more. Figure 1 show that the RBV dose per kg is generally lower with increasing weight.
Suppression of HCV RNA continued to be effective in both patient groups. Thus, EVR (92.0 vs. 83.3%, P=0.634), SVR (82.6 vs. 73.1%, P=0.506) and recurrence rates (4.3 vs. 3.8%, P=1.000) were not significantly different between Group A and Group B (Table 2).
Adverse events were reported by 79.6% (39/49) of patients (Table 3). Adverse events included flu-like symptoms, anemia, neutropenia, nausea, itching sensation, rash, thyroid dysfunction, depression, insomnia and thrombocytopenia. Flu-like symptoms were the most frequently reported adverse events (46.9%) and anemia was the most frequent reason for dose modification because of an adverse event (38.8%). Significant anemia (i.e. hemoglobin levels decreased to less than 10 g/dL) occurred more frequently in the Group A, but occurrence of other adverse events was similar between the two treatment groups (60.9 vs. 26.9%, P=0.022).
The reasons for RBV dose reduction were identified for all patients who completed treatment. Among the patients who completed 48 weeks of treatment, 20 patients (14 patients in Group A and 6 patients in Group B) had RBV dose reductions for safety reasons, including both adverse events (flu-like symptoms) and laboratory abnormalities (neutropenia and anemia). The RBV dose reduction rate was significantly higher for Group A than for Group B (60.9 vs. 23.1%) (P=0.01).
In Korea, a high endemic area for hepatitis B virus infection, about five to nine hundred thousand people are chronically infected with HCV, and at least 10-17% of these people get the hepatocellular carcinoma.10,11 HCV genotypes 1b and 2a are the two most common types, present in almost equal proportions in Korea.12,13 The current recommended therapy for CHC is the combination of Peg-IFN alfa and RBV. Although there are some guidelines, there continues to be debate regarding the optimal RBV dose.
In this report we present a detailed analysis of the impact of RBV dose reductions on SVR rates in patients naive to treatment, infected with HCV genotype 1 and treated with Peg-IFN α-2b and RBV combination therapy. The European Association for the Study of the Liver (EASL) clinical practice guidelines recommended that Peg-IFN α-2b should be used at a weight-based dose of 1.5 µg/kg per week and patients infected with HCV genotype 1 should receive the weight-based dose of RBV of 15 mg/kg body weight per day. The choice of this regimen was based upon the results of three pivotal, randomized, clinical trials that demonstrated the superiority of this combination treatment over standard IFN alfa and RBV combination therapy.14-16 Although RBV had only a modest transient effect on HCV clearance in the absence of IFN,17 it greatly enhanced SVR rates when given in combination with IFN alpha.18-20 In patients treated with Peg-IFN, RBV significantly accelerated the second/third phase of HCV clearance and treatment with Peg-IFN combined with RVB enhanced the rates of both EVR and SVR relative to that of Peg-IFN alone.21,22 The synergy between RBV and Peg-IFN can double the SVR rate obtained with Peg-IFN monotherapy. RBV acts by sustaining the virological response to Peg-IFN alpha.23 However, RBV treatment causes several side effects, including anemia, myalgia, flu-like symptoms and laboratory abnormalities. Among the side effects associated with RBV, anemia poses the most challenging side effect to manage and the degree of anemia has been correlated to plasma RBV levels. Anemia was also more frequent and serious in high-dose RBV treated patients.24 As a result, it is important to determine the optimal RBV dose that will maintain appropriate SVR, while decreasing its side effects. In some cases, significant anemia may lead to dose reduction and treatment discontinuation, thereby resulting in suboptimal outcomes. The relationship between hemoglobin decline and improved treatment response was noted from previous studies.25,26 In patients with HCV genotype 1 infection who developed anemia or who experienced a decline in hemoglobin >3.0 g/dL, even if they did not become anemic, these studies have shown that the odds of achieving SVR were approximately twofold increased, compared to patients who did not develop similar hematological changes. This relationship was identified with or without the inclusion of patients who received erythropoietin.27 Hematopoietic growth factors, such as erythropoietin, have been used to maintain effective hemoglobin concentrations during antiviral therapy. These growth factors have been shown to improve the quality of life, but not SVR rates.
In the Korean Association for the Study of the Liver (KASL) guidelines, weight-based RBV (1,000 mg for patients weighing <75 kg or 1,200 mg for patients weighing ≥75 kg) combined with Peg-IFN α-2b therapy was recommended for patients with genotype 1 CHC. A previous Korean report showed combination therapy efficacy comparable to that of studies in Western countries. Dosage modification was performed in 39% (29/75) of patients without producing a significant reduction in SVR.28
It has been known that the mean body weight of patients in Korea is relatively lower than in the western countries. Based on the current Korean guidelines, it is possible that the recommended dose of RBV may be too high in low-weight patients, resulting in more frequent dose reduction due to adverse events. The aim of this study was therefore to evaluate the possibility of an over-dose RBV treatment when we treat the patients according to the current Korean guidelines and to evaluate whether relatively high-dose RBV (≥15 mg/kg/day) causes RBV dose reduction more frequently. In our study, we referred to the EASL guideline,29 because the RBV dose of most patients does not exceed 15 mg/kg/day according to this guideline. We defined a high-dose RBV group (Group A) as a group of patients prescribed an initial RBV dose of 15 mg/kg/day or more. According to the KASL guideline, we treated patients with Peg-IFN α-2b (1.5 µg/kg per week s.c.) and RBV based on body weight (1,000 mg for patients weighing <75 kg or 1,200 mg for patients weighing ≥75 kg). In this analysis, we focused primarily on the RBV dose reduction rate because more than half of the patients (60.9%) who received more than 15 mg/kg/day of RBV as their initial dose experienced RBV dose reductions.
In order to minimize the effect of Peg-IFN, we included only one kind of Peg-IFN, which is Peg-IFN α-2b, and limited the cases to those administered for over 48 weeks. A randomized trial determined that the optimal duration of treatment should be based on the viral genotype and established that patients with genotype 1 should be treated for 48 weeks with Peg-IFN alfa plus standard weight-based RBV.16 For patients infected with HCV genotype 1, a higher dosage of RBV (i.e. 1,000 to 1,200 mg/day) and a treatment duration of at least 48 weeks were necessary to achieve an SVR rate of approximately 50%.16 Thus, patients infected with HCV genotype 1 are considered more difficult to treat, and intensive treatment regimens have been therefore suggested to maximize the virologic response.24 For most therapeutic regimens, treatment success is highly correlated with drug exposure. This relationship, however, is less well understood for patients with HCV infection. There is some evidence that dose reductions could negatively affect SVR rate in patients with genotype 1 HCV infection.30 In contrast, another study has shown that reductions in RBV dose after week 20, or Peg-IFN alfa-2a dose reductions throughout 48 weeks of treatment, had no effect on SVR rates.31
Dose reductions were less common in patients who received less than 15 mg/kg/day of RBV as their initial dose (23.1%). According to our findings, high-dose RBV (>15 mg/kg/day) appeared not to increase the virologic response, but rather prompted dose reduction more frequently (60.9%, 14/23). SVR was not significantly different between the two groups, but the dose reduction rate for anemia was significantly higher in the group administered at a dose of over 15 mg/kg/day. Most of the enrolled patients in our study (81.6%) weighed less than 70 kg, and half of these patients weighed 60 kg or less. Figure 1 show that relatively higher RBV doses (≥15 mg/kg/day) were administered frequently to the patients who had a lower body weight. According to the EASL guideline, most patients have to receive initial RBV doses as 15 mg/kg/day. Compared to this, the Korean guideline suggests over 15 mg/kg/day for low-weight patients and this has the risk of unnecessarily excessive administration of RBV. Accordingly, while the current Korean guideline suggests 1,200 mg for patients over 75 kg and 1,000 mg for those under 75 kg, it is considered necessary to add a new standard of RBV administration that applies less than 1,000 mg to low-weight patients who satisfy specific conditions or to establish a new guideline similar to the EASL guideline for the administration of RBV per kg suitable to Koreans.
There were some limitations in this study. First, this study was a retrospective study and examined only a small number of patients. Second, although we included patients who received Peg-IFN α-2b over 48 weeks in this study, we may have overlooked the impact of the total dose of Peg-IFN α-2b during therapy. Third, there was no evidence to support that treatment withdrawal or serious side effects frequently occurred in high-dose RBV treated patients (Group A) compared to low-dose RBV treated patients (Group B). Fourth, RBV bioavailability displays wide inter-individual variability and its plasma concentrations correlate poorly with the daily RBV dose. After adjustment for body weight, therapeutic drug monitoring of RBV has been considered a feasible option for improving RBV dose titrations.32,33 Because we did not use this monitoring option, we could not estimate the administered RBV dose accurately. Fifth, since other studies had offered conflicting results that administered total RBV dose during treatment associated with the SVR, this cannot be concluded from one-sided view. Finally, because mean body weight of woman is relatively lower compared to man, more women were included to the Group A and this fact could influence to this study results, especially SVR.
An important part of this study was to assess the RBV dose effect on the response to combination treatment with Peg-IFN alfa-2b. Based on our findings, we suggest that a lower dose of RBV than that proposed by the Korean standard guideline may be sufficiently effective and may decrease the need for dose reductions during treatment. However, because lowering the RBV dose can potentially decrease the SVR, further studies are needed to determine the optimal initial RBV dose required to maintain a virological response in genotype 1 chronic hepatitis C in Korean patients.
REFERENCES
2. Jacobson IM, Brown RS Jr, Freilich B, Afdhal N, Kwo PY, Santoro J, et al. Peginterferon alfa-2b and weight-based or flat-dose ribavirin in chronic hepatitis C patients: a randomized trial. Hepatology 2007;46:971-981. 17894303.


3. Rodriguez-Torres M, Jeffers LJ, Sheikh MY, Rossaro L, Ankoma-Sey V, Hamzeh FM, et al. Peginterferon alfa-2a and ribavirin in Latino and non-Latino whites with hepatitis C. N Engl J Med 2009;360:257-267. 19144941.


4. Hepburn MJ, Hepburn LM, Cantu NS, Lapeer MG, Lawitz EJ. Differences in treatment outcome for hepatitis C among ethnic groups. Am J Med 2004;117:163-168. 15276594.


5. Missiha S, Heathcote J, Arenovich T, Khan K. Canadian Pegasys Expanded Access Group. Impact of asian race on response to combination therapy with peginterferon alfa-2a and ribavirin in chronic hepatitis C. Am J Gastroenterol 2007;102:2181-2188. 17640318.


6. Kim KT, Han SY, Kim JH, Yoon HA, Baek YH, Kim MJ, et al. Clinical outcome of pegylated interferon and ribavirin therapy for chronic hepatitis C. Korean J Hepatol 2008;14:36-45. 18367856.


7. Lee HJ, Eun JR, Choi JW, Kim KO, Moon HJ. Comparison of therapeutic results between combination therapy of peginterferon alpha 2a plus ribavirin and interferon alpha-2b plus ribavirin according to treatment duration in patients with chronic hepatitis C. Korean J Hepatol 2008;14:46-57. 18367857.


8. Kang MJ, Jung EU, Park SW, Choi P, Kim JH, Park SJ, et al. Effects of pegylated interferon and ribavirin in Korean patients with chronic hepatitis C virus infection. Korean J Hepatol 2008;14:318-330. 18815455.


9. Reddy KR, Shiffman ML, Morgan TR, Zeuzem S, Hadziyannis S, Hamzeh FM, et al. Impact of ribavirin dose reductions in hepatitis C virus genotype 1 patients completing peginterferon alfa-2a/ribavirin treatment. Clin Gastroenterol Hepatol 2007;5:124-129. 17196435.


10. Huh K, Choi SY, Whang YS, Lee DS. Prevalence of viral hepatitis markers in Korean patients with hepatocellular carcinoma. J Korean Med Sci 1998;13:306-310. 9681811.



11. Lim YS. Current status of liver disease in Korea: hepatitis C. Korean J Hepatol 2009;15(Suppl 6):S25-S28. 20037276.


13. Kim YS, Ahn YO, Lee HS. Risk factors for hepatitis C virus infection among Koreans according to the hepatitis C virus genotype. J Korean Med Sci 2002;17:187-192. 11961301.



14. Manns MP, McHutchison JG, Gordon SC, Rustgi VK, Shiffman M, Reindollar R, et al. Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: a randomised trial. Lancet 2001;358:958-965. 11583749.


15. Fried MW, Shiffman ML, Reddy KR, Smith C, Marinos G, Gonçales FL Jr, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med 2002;347:975-982. 12324553.


16. Hadziyannis SJ, Sette H Jr, Morgan TR, Balan V, Diago M, Marcellin P, et al. Peginterferon-alpha2a and ribavirin combination therapy in chronic hepatitis C: a randomized study of treatment duration and ribavirin dose. Ann Intern Med 2004;140:346-355. 14996676.


17. Herrmann E, Lee JH, Marinos G, Modi M, Zeuzem S. Effect of ribavirin on hepatitis C viral kinetics in patients treated with pegylated interferon. Hepatology 2003;37:1351-1358. 12774014.


18. Pawlotsky JM, Dahari H, Neumann AU, Hezode C, Germanidis G, Lonjon I, et al. Antiviral action of ribavirin in chronic hepatitis C. Gastroenterology 2004;126:703-714. 14988824.


19. Bodenheimer HC Jr, Lindsay KL, Davis GL, Lewis JH, Thung SN, Seeff LB. Tolerance and efficacy of oral ribavirin treatment of chronic hepatitis C: a multicenter trial. Hepatology 1997;26:473-477. 9252161.


20. McHutchison JG, Gordon SC, Schiff ER, Shiffman ML, Lee WM, Rustgi VK, et al. Hepatitis Interventional Therapy Group. Interferon alfa-2b alone or in combination with ribavirin as initial treatment for chronic hepatitis C. N Engl J Med 1998;339:1485-1492. 9819446.


21. Poynard T, Marcellin P, Lee SS, Niederau C, Minuk GS, Ideo G, et al. International Hepatitis Interventional Therapy Group (IHIT). Randomised trial of interferon alpha2b plus ribavirin for 48 weeks or for 24 weeks versus interferon alpha2b plus placebo for 48 weeks for treatment of chronic infection with hepatitis C virus. Lancet 1998;352:1426-1432. 9807989.


22. Di Bisceglie AM, Hoofnagle JH. Optimal therapy of hepatitis C. Hepatology 2002;36(5 Suppl 1):S121-S127. 12407585.


23. Bronowicki JP, Ouzan D, Asselah T, Desmorat H, Zarski JP, Foucher J, et al. Effect of ribavirin in genotype 1 patients with hepatitis C responding to pegylated interferon alfa-2a plus ribavirin. Gastroenterology 2006;131:1040-1048. 17030174.


24. Lindahl K, Stahle L, Bruchfeld A, Schvarcz R. High-dose ribavirin in combination with standard dose peginterferon for treatment of patients with chronic hepatitis C. Hepatology 2005;41:275-279. 15660393.


25. Mac Nicholas R, Norris S. Review article: optimizing SVR and management of the haematological side effects of peginterferon/ribavirin antiviral therapy for HCV - the role of epoetin, G-CSF and novel agents. Aliment Pharmacol Ther 2010;31:929-937. 20175767.


26. Sulkowski MS, Shiffman ML, Afdhal NH, Reddy KR, McCone J, Lee WM, et al. Hepatitis C virus treatment-related anemia is associated with higher sustained virologic response rate. Gastroenterology 2010;139:1602-1611. 20723545.


27. Sievert W, Dore GJ, McCaughan GW, Yoshihara M, Crawford DH, Cheng W, et al. Virological response is associated with decline in hemoglobin concentration during pegylated interferon and ribavirin therapy in hepatitis C virus genotype 1. Hepatology 2011;53:1109-1117. 21480317.


28. Shiffman ML, Di Bisceglie AM, Lindsay KL, Morishima C, Wright EC, Everson GT, et al. Peginterferon alfa-2a and ribavirin in patients with chronic hepatitis C who have failed prior treatment. Gastroenterology 2004;126:1015-1023. 15057741.


29. Ghany MG, Strader DB, Thomas DL, Seeff LB. American Association for the Study of Liver Diseases. Diagnosis, management, and treatment of hepatitis C: an update. Hepatology 2009;49:1335-1374. 19330875.


30. McHutchison JG, Manns M, Patel K, Poynard T, Lindsay KL, Trepo C, et al. Adherence to combination therapy enhances sustained response in genotype-1-infected patients with chronic hepatitis C. Gastroenterology 2002;123:1061-1069. 12360468.


31. Lee H, Choi MS, Paik SW, Kim JH, Kim DY, Lee JH, et al. Peginterferon alfa-2a plus ribavirin for initial treatment of chronic hepatitis C in Korea. Korean J Hepatol 2006;12:31-40. 16565604.

Table 1.
Baseline clinical characteristics of the investigated patients
| Characteristic | Group A (RBV dose ≥15 mg/kg/day) | Group B (RBV dose <15 mg/kg/day) (N=26) | P-value |
|---|---|---|---|
| Mean age | 42.26 ±14.26 | 45.85 ±9.27 | 0.297* |
| Sex – no. (%) | |||
| Male | 7 (30.4) | 20 (76.9) | 0.020† |
| Female | 16 (69.6) | 6 (23.1) | |
| Body weight (kg) | 57.16 ±6.26 | 70.47 ±10.84 | 0.000* |
| BMI (kg/m2) | 21.84 ±1.70 | 25.05 ±2.91 | 0.000* |
| ALT, IU/L | 81.48 ±100.78 | 138.08 ±201.52 | 0.229* |
| HCV RNA, ×105 | 39.67 ±54.30 | 52.21 ±61.63 | 0.456* |
| Initial RBV dose (mg/kg/day) | 17.22 ±1.6 | 12.72 ±1.76 | 0.000* |
Table 2.
Clinical outcomes after treatment with combination therapy
| Group A (n=23) | Group B (n=26) | P-value* | |
|---|---|---|---|
| EVR rate | 21 (91.3) | 22 (84.6) | 0.634 |
| SVR rate | 19 (82.6) | 19 (73.1) | 0.506 |
| Recurrence rate | 1 (4.3) | 1 (3.8) | 1.000 |
| Dose reduction rate | 14 (60.9) | 6 (23.1) | 0.010 |
Table 3.
Frequencies of adverse events and dose modifications
| Adverse events |
Group A |
Group B |
P-value* | ||
|---|---|---|---|---|---|
| n (%) | Dose modification, n (%) | n (%) | Dose modification, n (%) | ||
| Flu-like symptoms | 14 (28.6) | 4 (8.2) | 9 (18.4) | 3 (6.1) | 0.352 |
| Anemia | 14 (28.6) | 13 (26.5) | 7 (14.3) | 6 (12.2) | 0.022 |
| Neutropenia | 5 (10.2) | 3 (6.1) | 11 (22.4) | 3 (6.1) | 1.000 |
| Nausea | 4 (8.2) | 0 (0.0) | 6 (12.2) | 0 (0.0) | 0.731 |
| Itching sensation | 2 (4.1) | 0 (0.0) | 6 (12.2) | 0 (0.0) | 0.254 |
| Rash | 2 (4.1) | 1 (2.0) | 4 (8.2) | 0 (0.0) | 0.671 |
| Thyroid dysfunction | 2 (4.1) | 0 (0.0) | 4 (8.2) | 0 (0.0) | 1.000 |
| Depression | 2 (4.1) | 0 (0.0) | 2 (4.1) | 0 (0.0) | 0.215 |
| Insomnia | 2 (4.1) | 0 (0.0) | 2 (4.1) | 0 (0.0) | 0.215 |
| Thrombocytopenia | 2 (4.1) | 0 (0.0) | 1 (2.0) | 0 (0.0) | 0.469 |



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




