| Clin Mol Hepatol > Volume 28(1); 2022 > Article |
|
ABSTRACT
Background/Aims
Methods
Results
ACKNOWLEDGMENTS
FOOTNOTES
SUPPLEMENTAL MATERIAL
Supplementary┬ĀTable┬Ā1.
Supplementary┬ĀTable┬Ā2.
Supplementary┬ĀTable┬Ā3.
Supplementary┬ĀTable┬Ā5.
Supplementary┬ĀTable┬Ā6.
Supplementary┬ĀFigure┬Ā1.
Supplementary┬ĀFigure┬Ā2.
Supplementary┬ĀFigure┬Ā3.
Supplementary┬ĀFigure┬Ā4.
Supplementary┬ĀFigure┬Ā5.
Supplementary┬ĀFigure┬Ā6.
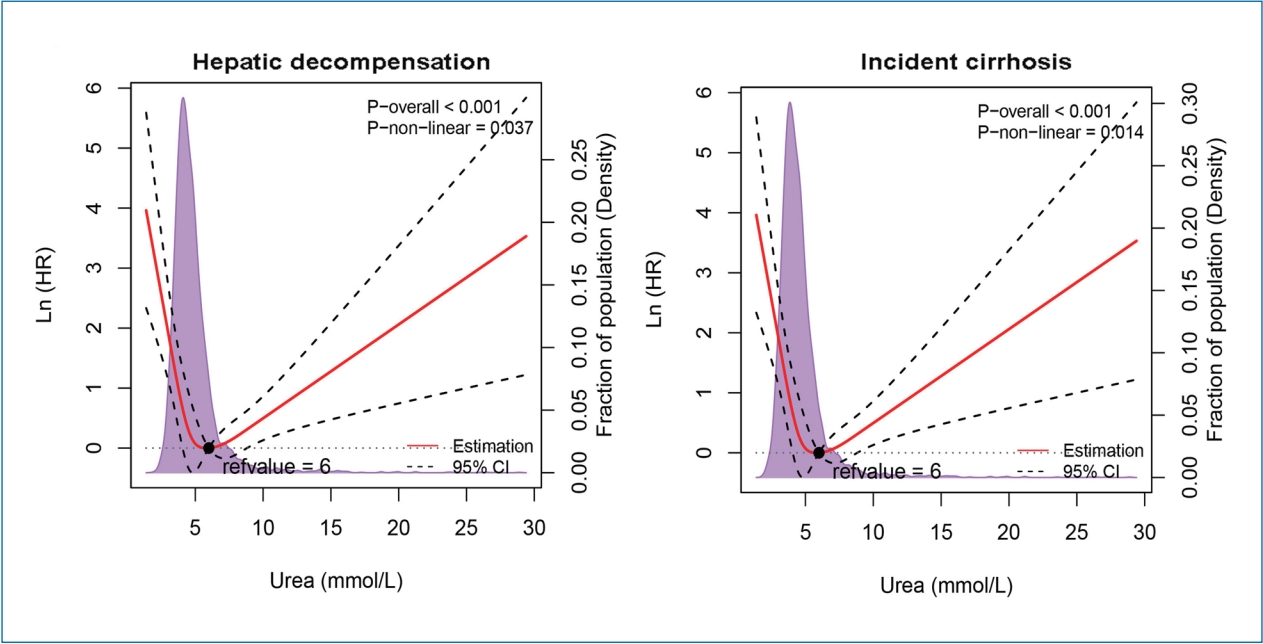
Figure┬Ā2.
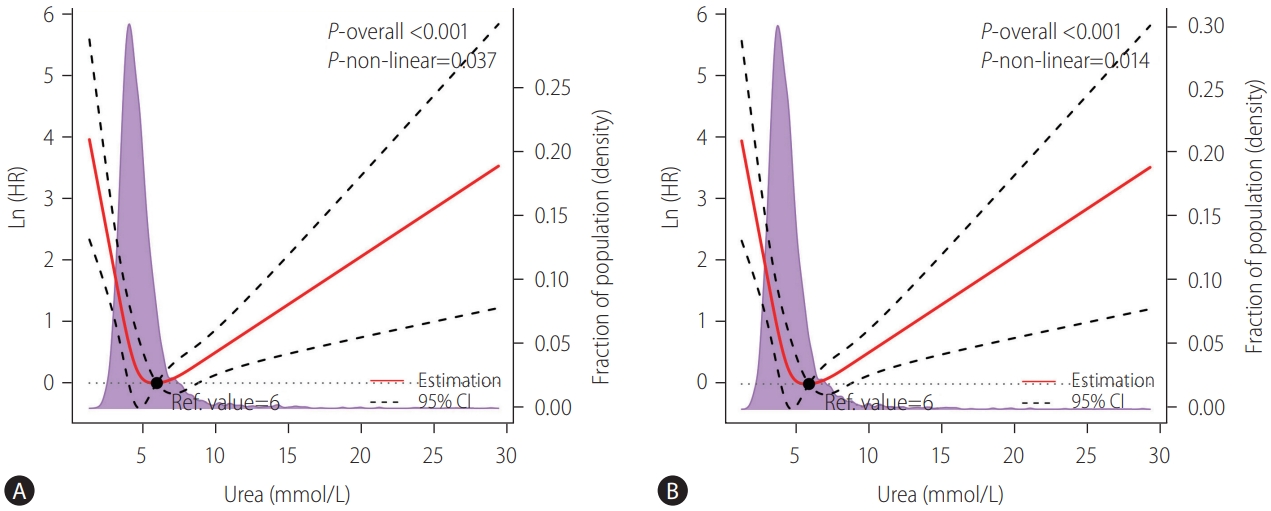
Figure┬Ā3.
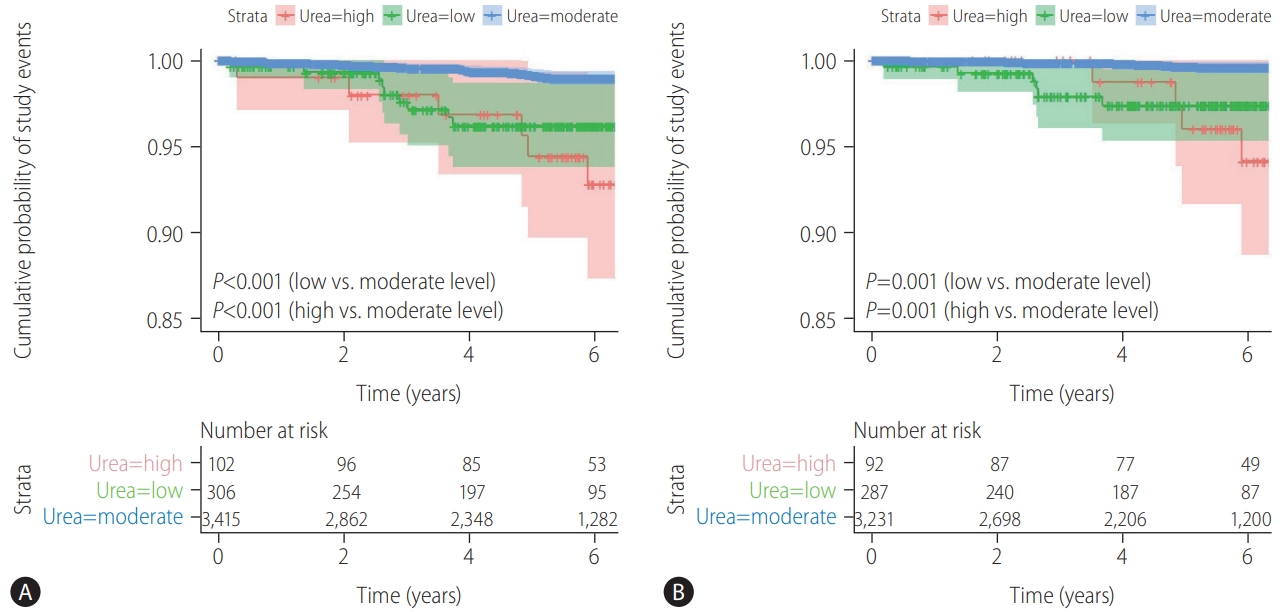
Figure┬Ā4.
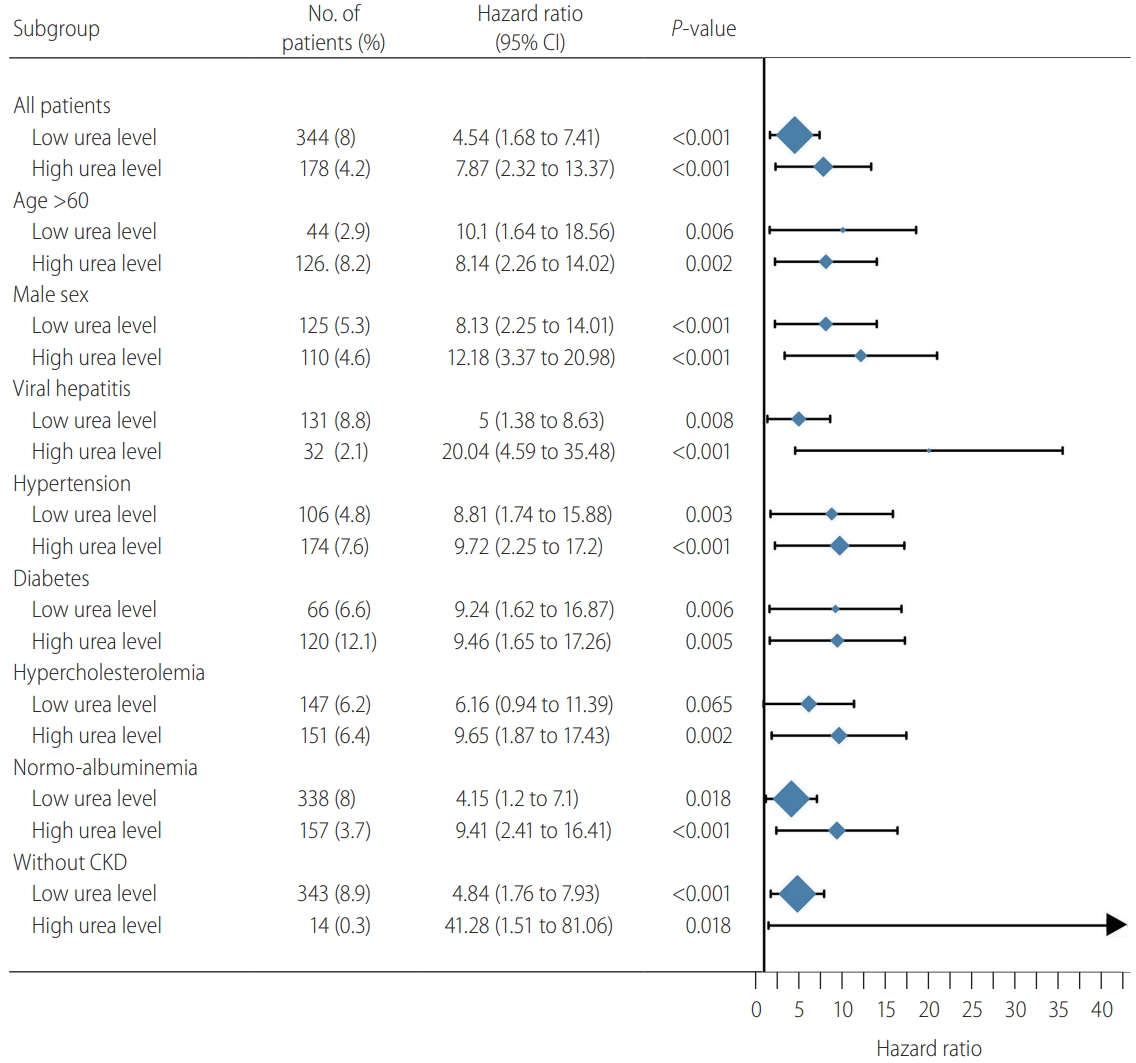
Figure┬Ā5.
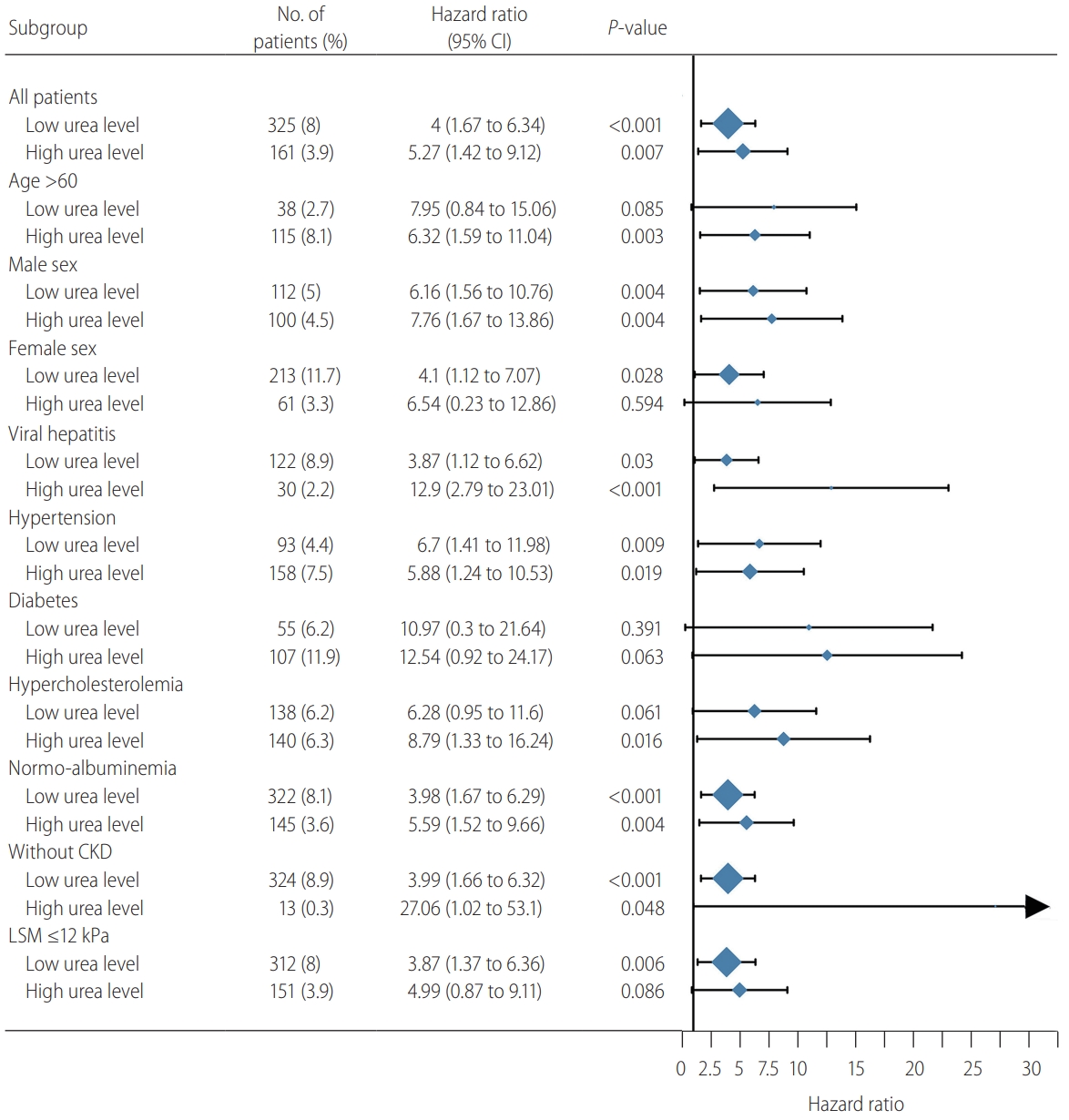
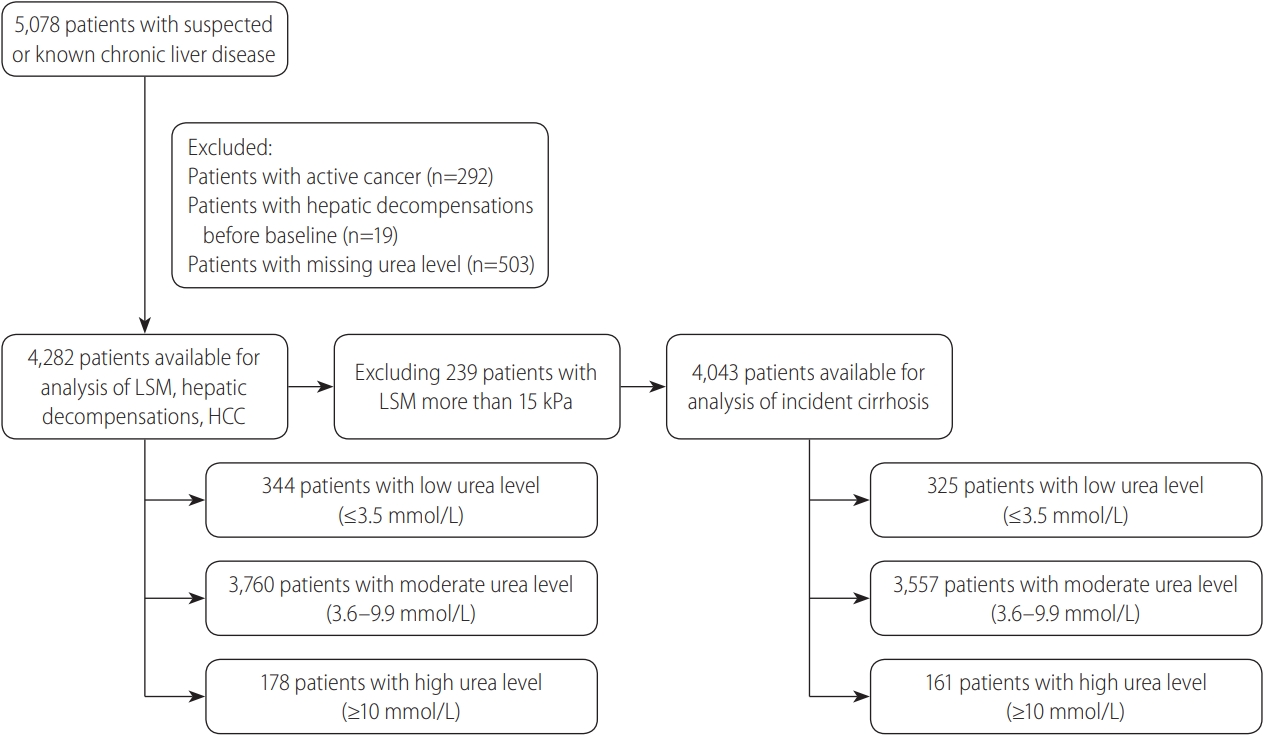
Table┬Ā1.
| Characteristic | All | Low urea level | Moderate urea level | High urea level | P-value |
|---|---|---|---|---|---|
| Total | 4,282 | 344 | 3,760 | 178 | |
| Age (years) | 55.6┬▒12.3 | 47.7┬▒11.5 | 55.9┬▒12.0 | 65.6┬▒9.9 | <0.001 |
| Male sex | 2,367 (55.3) | 125 (36.3) | 2,132 (56.7) | 110 (61.8) | <0.001 |
| HBV infection | 1,375 (32.1) | 125 (36.3) | 1,218 (32.4) | 32 (18.0) | <0.001 |
| HCV infection | 131 (3.1) | 6 (1.7) | 124 (3.3) | 1 (0.6) | 0.039 |
| NAFLD | 1,581 (36.9) | 106 (30.8) | 1,410 (37.5) | 65 (36.5) | 0.048 |
| Type 2 diabetes mellitus | 994 (23.2) | 66 (19.2) | 808 (21.5) | 120 (67.4) | <0.001 |
| Hypertension | 2,290 (53.5) | 109 (31.7) | 2007 (53.4) | 151 (84.8) | <0.001 |
| Hypercholesterolemia | 2364 (55.2) | 147 (42.7) | 2066 (54.9) | 151 (84.8) | <0.001 |
| Body mass index (kg/m2) | 26 (23ŌĆō28) | 25 (22ŌĆō28) | 26 (23ŌĆō28) | 27 (24ŌĆō29) | 0.021 |
| ALT (IU/L) | 26 (18ŌĆō39) | 26 (16ŌĆō44) | 26 (19ŌĆō39) | 21 (16ŌĆō29) | <0.001 |
| Albumin (g/L) | 43.6┬▒3.1 | 43.4┬▒3.4 | 43.8┬▒3.0 | 40.2┬▒3.8 | <0.001 |
| Total bilirubin (╬╝mol/L) | 10.5 (7.9ŌĆō14.0) | 10.3 (7.5ŌĆō14.2) | 10.6 (8.1ŌĆō14.1) | 7.1 (5.6ŌĆō10.0) | <0.001 |
| ALP (IU/L) | 67 (56ŌĆō82) | 66 (53ŌĆō79) | 67 (56ŌĆō81) | 76 (62ŌĆō91) | <0.001 |
| AFP (╬╝g/L) | 2.8 (1.9ŌĆō4.1) | 2.7 (1.8ŌĆō4.3) | 2.8 (2.0ŌĆō4.1) | 2.0 (1.5ŌĆō2.8) | 0.034 |
| WCC (├Ś109/L) | 6.6┬▒1.9 | 6.4┬▒2.1 | 6.6┬▒1.9 | 7.4┬▒1.9 | <0.001 |
| Platelet (├Ś109/L) | 214 (176ŌĆō255) | 229 (188ŌĆō276) | 213 (174ŌĆō253) | 214 (183ŌĆō252) | <0.001 |
| Prothrombin time (seconds) | 10.7 (10.1ŌĆō11.3) | 10.7 (10.3ŌĆō11.3) | 10.7 (10.1ŌĆō11.3) | 10.4 (9.8ŌĆō11.1) | 0.054 |
| INR | 1.0 (0.9ŌĆō1.1) | 1.0 (1.0ŌĆō1.0) | 1.0 (0.9ŌĆō1.0) | 1.0 (0.9ŌĆō1.0) | 0.079 |
| Triglycerides (mmol/L) | 1.2 (0.8ŌĆō1.7) | 1.1 (0.8ŌĆō1.5) | 1.2 (0.8ŌĆō1.7) | 1.5 (1.0ŌĆō2.4) | <0.001 |
| Cholesterol (mmol/L) | 4.5 (3.9ŌĆō5.2) | 4.7 (4.0ŌĆō5.3) | 4.5 (3.9ŌĆō5.2) | 4.3 (3.7ŌĆō4.8) | <0.001 |
| FBS (mmol/L) | 6.0 (5.1ŌĆō7.7) | 5.3 (4.8ŌĆō7.0) | 6.0 (5.1ŌĆō7.7) | 6.9 (5.6ŌĆō8.5) | <0.001 |
| Haemoglobin A1c (%) | 6.7 (5.8ŌĆō7.8) | 6.1 (5.6ŌĆō7.5) | 6.7 (5.8ŌĆō7.7) | 7.6 (6.8ŌĆō8.7) | <0.001 |
| Positive HBeAg* | 213 (19.1) | 27 (24.5) | 184 (18.6) | 2 (12.5) | 0.253 |
| HBV DNA* (log10 IU/mL) | 4 (3ŌĆō6) | 4 (3ŌĆō6) | 4 (3ŌĆō6) | 2 (1-5) | 0.554 |
| Creatinine (┬Ąmol/L) | 75 (62ŌĆō89) | 62 (54ŌĆō72) | 75 (63ŌĆō88) | 184 (132ŌĆō256) | <0.001 |
| eGFR (mL/min/1.73 m2) | 92.7 (76.9ŌĆō104.2) | 107.2 (97.8ŌĆō115.5) | 92.3 (78.1ŌĆō103.2) | 29.0 (19.2ŌĆō42.5) | <0.001 |
| Urea (mmol/L) | 5.1 (4.3ŌĆō6.2) | 3.2 (2.9ŌĆō3.4) | 5.2 (4.4ŌĆō6.2) | 12.9 (10.9ŌĆō16.9) | <0.001 |
| LSM (kPa) | 5.9 (4.5ŌĆō7.9) | 5.4 (4.3ŌĆō7.2) | 5.9 (4.5ŌĆō7.9) | 6.3 (4.9ŌĆō9.0) | <0.001 |
| CAP (dB/m) | 262 (217ŌĆō310) | 255.5 (204.0ŌĆō303.5) | 263.0 (218.0ŌĆō311.0) | 254.0 (209.0ŌĆō309.5) | 0.028 |
| Medication use | |||||
| ŌĆāStatins | 1,807 (42.2) | 88 (25.6) | 1575 (41.9) | 144 (80.9) | <0.001 |
| ŌĆāAnti-platelet drugs | 834 (19.5) | 36 (10.5) | 704 (18.7) | 94 (52.8) | <0.001 |
| ŌĆāAntiviral treatment | 625 (14.6) | 48 (14.0) | 562 (14.9) | 15 (8.4) | 0.052 |
Values are presented as mean┬▒standard deviation, median (interquartile range), or number (%).
LSM, liver stiffness measurement; HBV, hepatitis B virus; HCV, hepatitis C virus; NAFLD, nonalcoholic fatty liver disease; ALT, alanine aminotransferase; ALP, alkaline phosphatase; AFP, alpha-fetoprotein; WCC, white cell count; INR, international normalized ratio; FBS, fasting blood sugar level; HBeAg, hepatitis B e antigen; eGFR, estimated glomerular filtration rate; CAP, controlled attenuation parameter.
Table┬Ā2.
Table┬Ā3.
Abbreviations
REFERENCES
- TOOLS
-
METRICS

- ORCID iDs
-
Vincent Wai-Sun Wong

https://orcid.org/0000-0003-2215-9410 - Related articles
-
Interaction between sarcopenia and nonalcoholic fatty liver disease2023 February;29(Suppl)
Microbiome and metabolomics in alcoholic liver disease2022 July;28(3)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Supplement1
Supplement1 Print
Print



