| Clin Mol Hepatol > Volume 30(2); 2024 > Article |
|
ABSTRACT
Background/Aims
Methods
Results
ACKNOWLEDGMENTS
FOOTNOTES
SUPPLEMENTAL MATERIAL
Supplementary┬ĀTable┬Ā1.
Supplementary┬ĀTable┬Ā2.
Supplementary┬ĀTable┬Ā3.
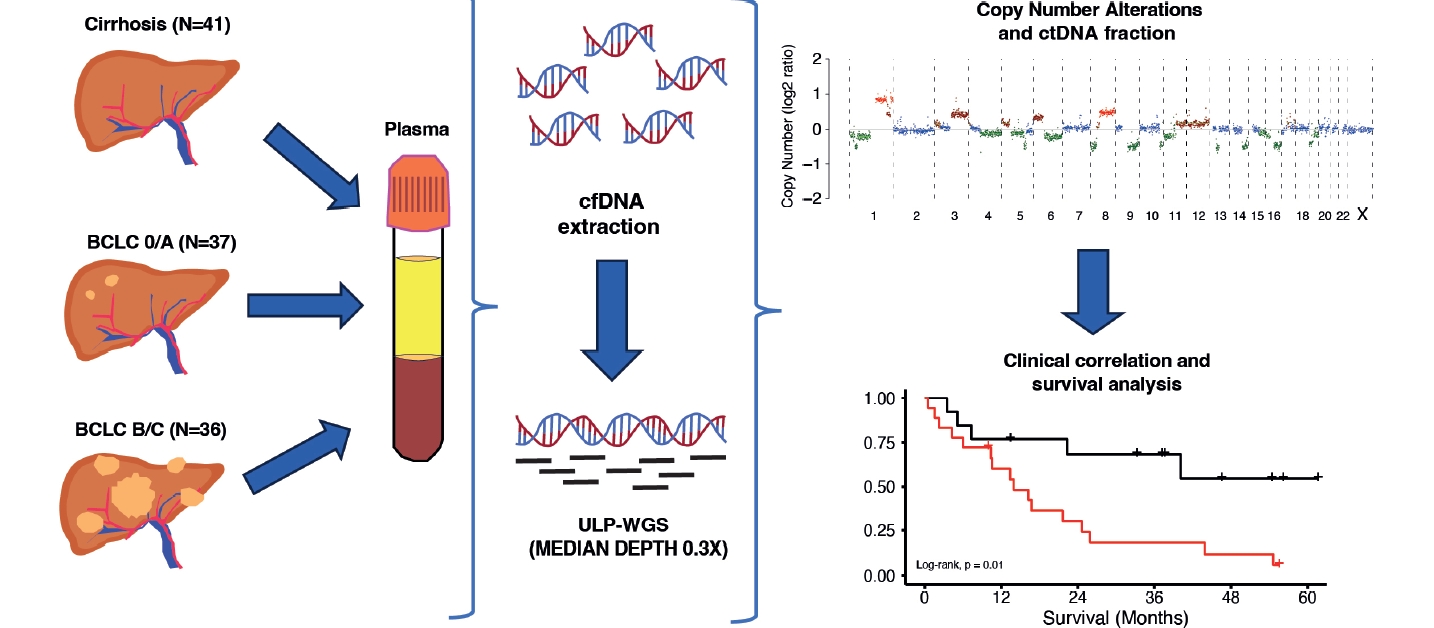
Figure┬Ā1.
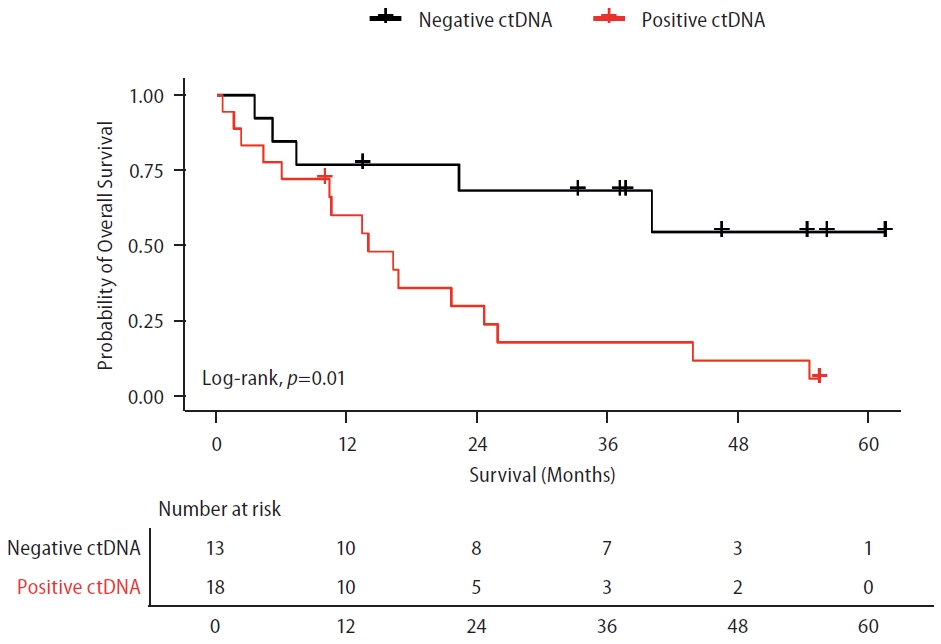
Figure┬Ā2.
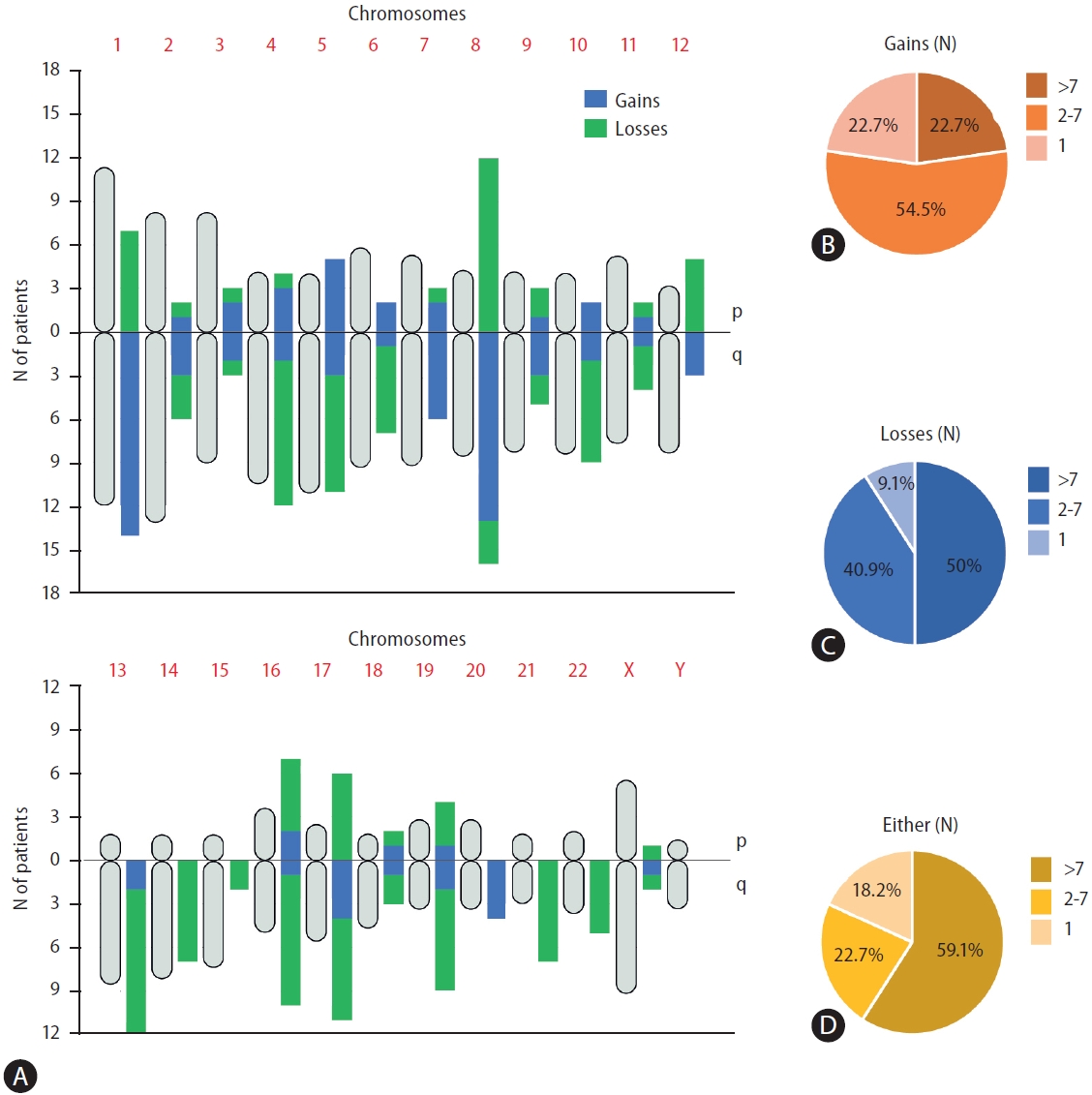
Figure┬Ā3.
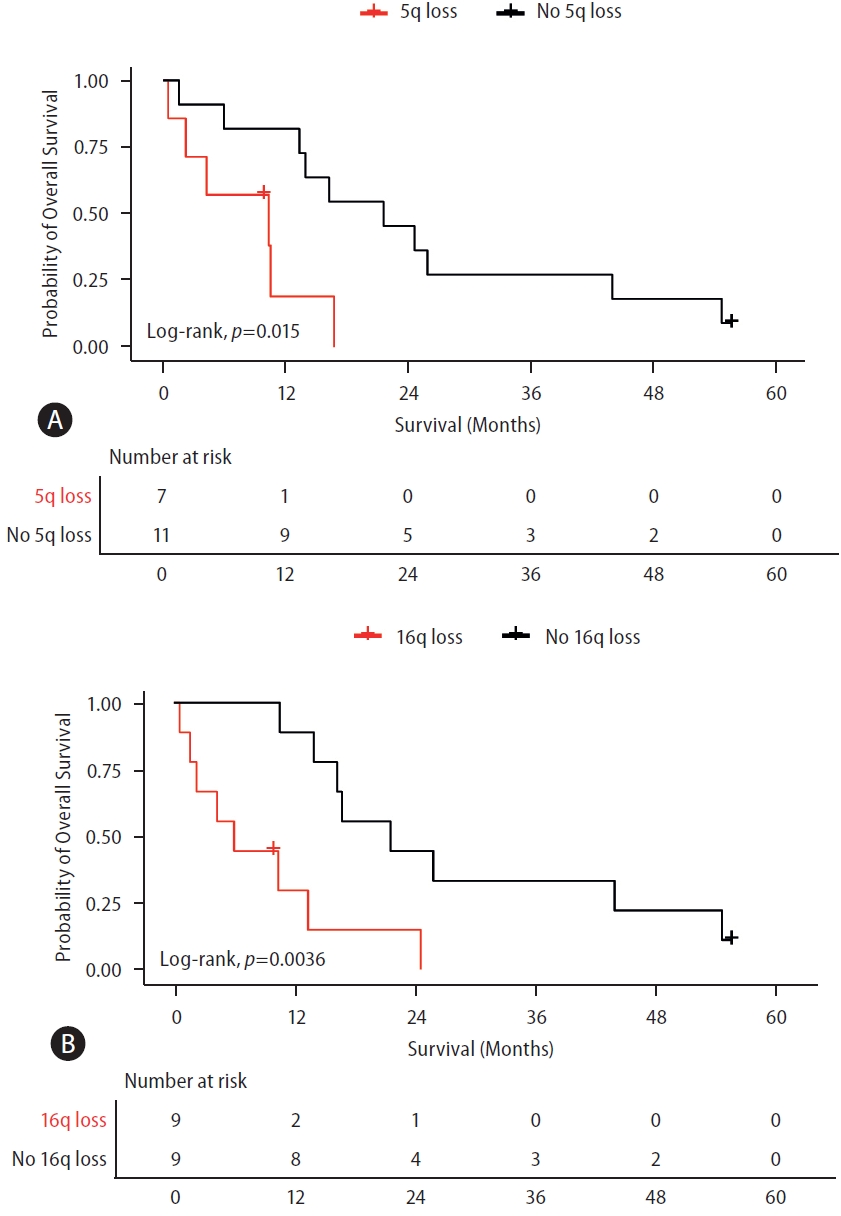
Table┬Ā1.
Values are presented as number only, number (%), or median (interquartile range).
HCC, hepatocellular carcinoma; HCV, hepatitis C virus; HBV, hepatitis B virus; MAFLD, metabolic dysfunction-associated fatty liver disease; ALBI, albumin-bilirubin; BCLC, Barcelona Clinic Liver Cancer; Anti-PD1, anti-programmed cell death protein 1; Anti-CTLA4, cytotoxic T lymphocyte-associated antigen 4; Anti-PD-L1, anti-programmed death-ligand 1; Anti-VEGF, anti-vascular endothelial growth factor.
Table┬Ā2.
Table┬Ā3.
| Clinical features | Positive ctDNA | Negative ctDNA | P-value |
|---|---|---|---|
| Number of patients | 22 | 51 | |
| Age, years | 63.68 (59.45-71.11) | 65.6 (59.9-69.77) | 0.491 |
| Male | 20 (90.90) | 46 (90.19) | 1 |
| Etiology | 0.802 | ||
| ŌĆāViral etiology | 8 (36.36) | 17 (33.33) | |
| ŌĆāNon-viral etiology | 14 (63.63) | 34 (66.67) | |
| BCLC stage | 0.000* | ||
| ŌĆā0/A | 3 (13.63) | 34 (66.67) | |
| ŌĆāB/C | 19 (86.36) | 17 (33.33) | |
| Macrovascular invasion | 0.023* | ||
| ŌĆāYes | 8 (36.36) | 6 (11.76) | |
| ŌĆāNo | 14 (63.63) | 45 (88.23) | |
| Extrahepatic spread | 0.000* | ||
| ŌĆāYes | 10 (45.45) | 4 (7.84) | |
| ŌĆāNo | 12 (54.54) | 47 (92.15) | |
| Bilobar involvement | 0.062 | ||
| ŌĆāYes | 11 (50) | 14 (27.45) | |
| ŌĆāNo | 11 (50) | 37 (72.54) | |
| Tumor number | 0.179 | ||
| ŌĆā<3 | 11 (50) | 34 (66.66) | |
| ŌĆāŌēź3 | 11 (50) | 17 (33.33) | |
| Tumor size | 0.000* | ||
| ŌĆā<5 cm | 10 (45.45) | 46 (90.19) | |
| ŌĆāŌēź5 cm | 12 (54.54) | 5 (9.80) | |
| AFP | 0.000* | ||
| ŌĆāŌēź20 ng/mL | 16 (72.72) | 9 (17.64) | |
| ŌĆā<20 ng/mL | 6 (27.27) | 42 (82.35) | |
| Treatments | 0.000* | ||
| Surgical | 2 (9.09) | 35 (68.62) | |
| ŌĆāLocoregional | 2 (9.09) | 3 (5.88) | |
| ŌĆāSystemic | 18 (81.81) | 13 (25.49) | |
| Systemic treatment | 0.718 | ||
| ŌĆāSorafenib | 11 (61.11) | 9 (69.23) | |
| ŌĆāImmunotherapy | 7 (38.88) | 4 (30.76) |
Abbreviations
REFERENCES
- TOOLS
-
METRICS

- ORCID iDs
-
Josepmaria Argemi

https://orcid.org/0000-0003-1696-7753 - Related articles
-
Current status of laparoscopic liver resection for hepatocellular carcinoma2016 June;22(2)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement1
Supplement1 Print
Print



