Mycophenolate mofetil as an alternative treatment for autoimmune hepatitis
Article information
Abstract
Autoimmune hepatitis (AIH) is an immune-mediated chronic liver disease characterized by hepatocellular inflammation, necrosis, and fibrosis, which can progress to cirrhosis and fulminant hepatic failure. The standard treatment for AIH includes corticosteroids alone or in combination with azathioprine. Although most patients achieve remission using the standard regimen, some patients do not respond due to either drug intolerance or refractory disease; in such cases alternative immunosuppressive agents should be explored. The second-line therapies are cyclophilin inhibitors such as cyclosporine A or tacrolimus, and nowadays mycophenolate mofetil (MMF) is widely used if azathioprine-based therapies are not tolerated. Although these are recommended as an alternative to the first-line regimen, there is insufficient evidence for the efficacy of second-line therapies, with the evidence based mainly on expert opinion. Therefore, we report an AIH patient receiving the standard regimen in whom remission did not occur due to side effects to azathioprine, but was successfully treated with MMF in combination with corticosteroids as an alternative to the standard regimen.
INTRODUCTION
Autoimmune hepatitis (AIH) is an immune-mediated chronic liver disease characterized by hepatocellular inflammation, necrosis, and fibrosis, which can progress to cirrhosis and fulminant hepatic failure if left untreated [1]. AIH has a female predominance occurring in all ages and races, and both the median and mean age of initial disease presentation are in the forties, similar to many other autoimmune diseases. Diagnostic criteria are based on demonstration of characteristic autoantibodies, elevated immunoglobulins, and histological features of hepatitis in the absence of viral liver disease [1]. Standard treatment of AIH includes corticosteroids alone or in combination with azathioprine (AZA) that aims at the normalization of transaminase and immunoglobulin G (IgG) levels in serum which correlate with histological disease activity [1,2]. It is reported that although majority of the patients respond well to the standard therapies, about 20% of patients do not due to either intolerance or refractory disease [2,3]. In treatment failure, alternative immunosuppressive and biological agents including mycophenolate mofetil (MMF), tacrolimus, cyclosporine and anti TNF antibodies should be considered [2]. The patient we report here is a 48-year old woman who was diagnosed with autoimmune hepatitis and was treated with standard therapy corticosteroids and AZA. However, she could not tolerate the side effect of AZA induced pancytopenia and skin rash. MMF in combination with corticosteroid was opted as second-line treatment and the patient was able to achieve remission with these treatments.
CASE REPORT
A 48 year-old woman was admitted to department of hepatology due to fatigue and jaundice which lasted for approximately 10 days. She also complained of anorexia, nausea and dark colored urination. She had history of heavy drinking (60 g of alcohol every day) and herbal tea intake (Solomon´s seal tea twice a week) for recent 6 months. On physical examination, icteric sclera was observed but there was neither hepatomegaly nor splenomegaly. Upon admission, the laboratory findings were as follows: alanine aminotransferase (ALT) level 472 IU/L, aspartate aminotransferase (AST) level 556 IU/L, alkaline phosphatase (ALP) 123 IU/L, total bilirubin 3.44 mg/dL, direct bilirubin 1.91 mg/dL and prothrombin time internal normalisation ratio (INR) 1.05. Viral markers including hepatitis A antibody immunoglobulin M (HAV Ab IgM), hepatitis B surface antigen (HBsAg) and hepatitis C antibody (HCV Ab) were negative. Since the patient was a middle aged woman, further assessments to rule out autoimmune hepatitis were also performed. Anti-smooth muscle antibody (SMA) and antinuclear antibody (ANA) were positive with the ratio of 1:640 for ANA and IgG was increased to 1,940 mg/dL. Anti-LKM and anti-mitochondrial antibody were negative. Liver biopsy was performed and the histologic findings revealed mild portal inflammation with lymphoplasmacytic, neutrophilic and eosinophilic infiltration along with minimal interface activity (Fig. 1). As the histologic findings were not typical for autoimmune hepatitis along with spontaneous slow decrease in AST (48 IU/L), ALT (49 IU/L) and total bilirubin (0.64 mg/dL) levels the patient was followed up as out-patient without treatment.

Microscopic findings showing mild portal inflammation (arrows) with lymphoplasmacytic, neutrophilic and eosinophilic infiltration along with is minimal interface activity (H & E stain, ×200).
Two months later, hepatitis flare recurred despite quitting alcohol and herbal tea. Follow up ANA was positive with the ratio of 1:1,280 and IgG level was further elevated to 2,420 mg/dL (Fig. 2A, B). Based on the lab findings and fluctuation of AST and ALT levels, diagnosis of autoimmune hepatitis type I was made. She was initially prescribed with prednisolone 30 mg which was tapered to 5 mg in in 4 months and was maintained in combination with AZA 25 mg daily. The patient showed treatment response with decrease in serum AST and ALT levels along with decrease in total IgG levels after 3 months of treatment (Fig. 2A, B). According to the autoimmune hepatitis scoring system [4], post-treatment scores was 18, which was compatible with definite AIH.
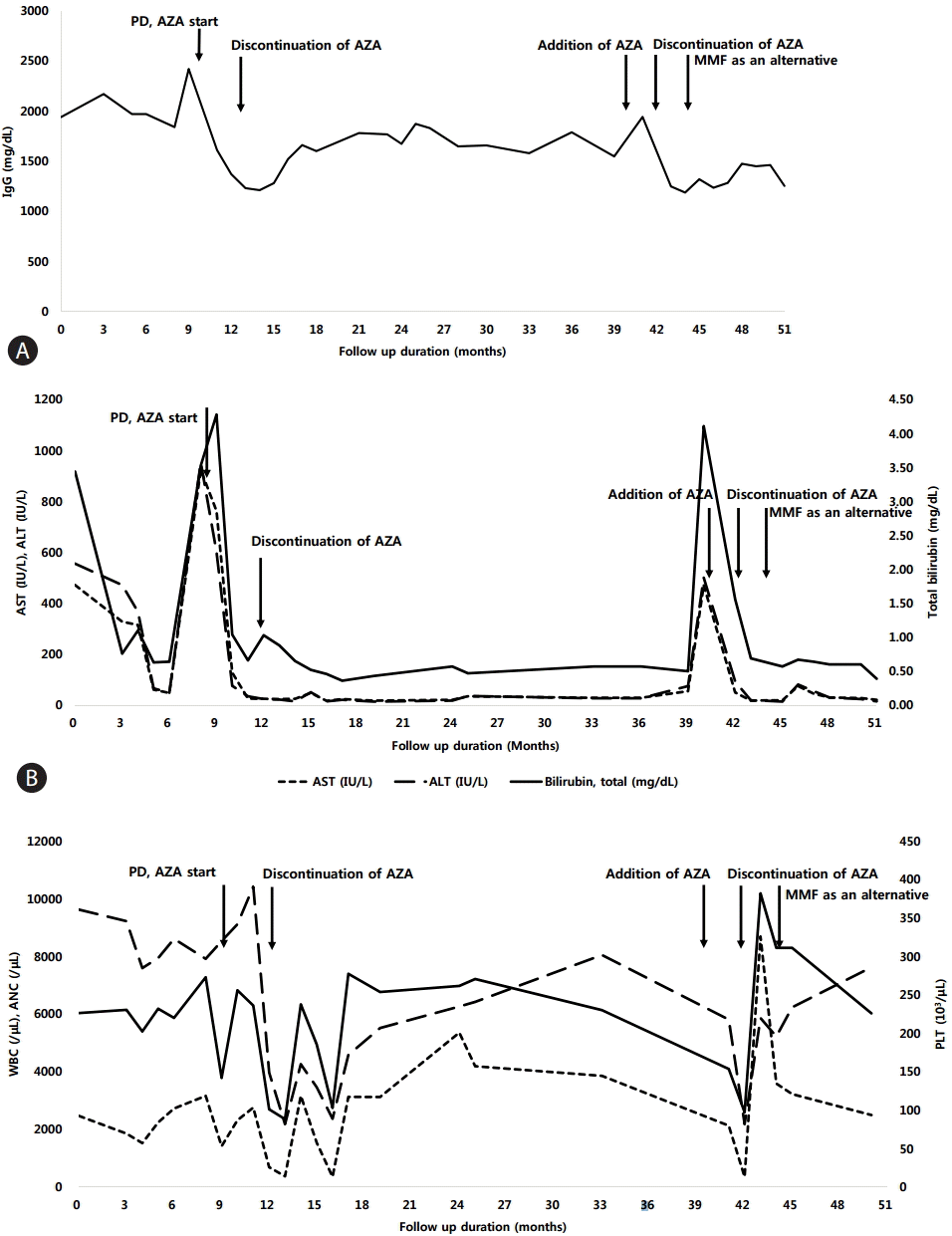
Changes in (A) serum total IgG, (B) liver enzymes and (C) complete blood cell count following treatment durations. The solid arrows indicate when each medication was started. PD, prednisolone; AZA, azathioprine; AST, aspartate aminotransferase; ALT, alanine aminotransferase; WBC, white blood cells; ANC, absolute neutrophil count; PLT, platelet; IgG, immunoglobulin G; ALP, alkaline phosphatase.
After 3 months of maintenance treatment, AZA-related side effect, pancytopenia and alopecia developed. Hemoglobin level decreased to 9.6 g/dL, white blood cell count decreased to 2,150 mm3/μL (absolute neutrophil count 377 mm3/μL) and platelet count decreased to 82,000 mm3/μL (Fig. 2C). Furthermore, hair loss occurred 2 months after treatment with AZA which aggravated until the patient had to wear a wig. Therefore, AZA was discontinued after 5 months of treatment and was maintained on 5mg of prednisolone daily. After discontinuation of AZA, complete blood count returned to normal range within 1 month while hair growth returned to normal 2 months later.
Although the symptoms subsided after quitting AZA, there were wax and wane in AST and ALT levels after 27 months of steroid monotherapy, exacerbation of AIH occurred. The steroid dose was escalated to 30 mg again in combination with 50 mg of azathioprine. The AST/ALT levels dropped to normal range after 20 days of treatment. Unfortunately, after 20 days of combination treatment, AZA-related side effect occurred again, especially severe alopecia was noted, unabling the patient to maintain on standard treatment. As remission was not achieved with first-line treatment, steroid monotherapy was maintained for another 2 months which was decreased to 15 mg during that duration and MMF 1 g per day was added thereafter. The steroid was tapered to 5 mg and MMF to 500 mg daily. After administration of MMF, her symptoms had subsided and normalization of liver enzymes and IgG level was achieved. Hair growth resumed and bone marrow suppression was improved. The patient is still on 5 mg of steroid and 500 mg of MMF and is stable for 12 months after the acute exacerbation of AIH.
DISCUSSION
Autoimmune hepatitis (AIH) was first reported in 1951 as a fluctuating persistent hepatitis of young woman with marked elevations of serum immunoglobulins [5,6]. The median and mean age of initial disease presentation are in the forties and the etiology of AIH is unknown, but it carries all features of an autoimmune disease: genetic predisposition, spontaneous disease fluctuations, circulating autoantibodies, auto-reactive T cells, inflammatory infiltrations in liver, and a good response to immunosuppressive agents [1,5,7-9]. AIH can be diagnosed when compatible clinical signs and symptoms, laboratory abnormalities (increased serum AST or ALT, and total IgG), serological (autoantibodies), and histological (interface hepatitis, lymphocytic and lymphoplasmacytic infiltrates in portal tracts and extending in the lobule, hepatic rosette formation, or chronic hepatitis with lymphocytic infiltration) findings are present; and other conditions that can cause chronic hepatitis, including viral, metabolic, cholestatic, hereditary, and drug-induced diseases, have been excluded [1].
Studies reported that when AIH is left untreated, as many as 40% of the patients with severe disease die within 6 months and of the remaining 40% develop cirrhosis [9,10]. Therefore, when clinical, laboratory or histologic features of active liver inflammation is present, immunosuppressive treatment should be initiated. Standard treatment includes corticosteroids alone or in combination with AZA that aims at the normalization of transaminase and IgG levels in serum which correlate with histological disease activity [1,8]. In adults with AIH, standard treatment can be started with prednisolone (starting with 30 mg daily and tapering down to 10 mg daily within 4 weeks) in combination with azathioprine (50 mg daily or 1-2 mg/kg body weight) or prednisolone alone (starting with 40-60 mg daily and tapering down to 20 mg daily within 4 weeks) [2].
Although most of the patients respond very well to immunosuppressive treatment, 20% of patients do not respond to or are intolerant to standard therapy [1,11]. The overall frequency of AZA-related side effect is 10%, which can be improved after dose reduction or discontinuation [12,13]. AZA is a non-selective immunosuppressant that acts by inhibition of several enzymes involved in purine synthesis and this contributes to side effects including bone marrow suppression, nausea, rash, alopecia, arthralgias, neoplastic and malabsorption. The frequency of cytopenia in azathioprine treated patients with autoimmune hepatitis is 46%, and the occurrence of severe hematologic abnormalities is 6% [2]. Thiopurine S-methyltransferase (TPMT) is part of a cascade of enzymes responsible for the metabolism of thioprine drugs including AZA, 6-mercaptopurine (6-MP) and 6 thioguanine (6-TG) [14]. Some studies reported that activity of TPMT could be used to predict those who would respond to treatment and those who would show hematologic side effects as the patients with deficient TPMT activity are at severe risk of developing bone marrow suppression [15-17]. However, TPMT testing is not routinely performed as it is time consuming and not widely available
In the past 15 years alternative immunosuppressive and biological agents for those who cannot tolerate standard treatment were evaluated. Unfortunately, the treatment options for patients who failed to respond to standard therapy is still limited. MMF, an ester pro-drug of mycophenolic acid which acts as a noncompetitive inhibitor of inosine monophosphate dehydrogenase, the rate-limiting enzyme involved in the de nevo synthesis of purines were studied as an alternative to standard treatment [18,19]. According to the AASLD practice guidelines, MMF or cyclosporine have had the most empiric use as alternative medications and MMF (2 g daily orally) is the most promising current agent [2] where improvement of hepatitis can be expected in 39-84% of patients.
Several studies suggested MMF as an alternative treatment for those who are either refractory or intolerant to steroid and azathioprine combination therapy [18,20,21]. Study by Zachou et al. [19] prospectively studied the efficacy and safety of MMF in treatment naïve AIH patients where 88% of 59 patients responded to the treatment and even allowed rapid steroid tapering with eventually withdrawal of steroid. Another study by Sharzehi et al. [20] based on retrospective cohort including 90 patients showed that those who were unable to continue corticosteroid and azathioprine well tolerated MMF with 88% of patients maintaining complete remission. These studies initiated MMF at a dose of 1.5-2 g/day in the former while 1g/day in the latter study. The reported side effect was gastrointestinal symptoms including nausea, vomiting and diarrhea, rash and hair loss but no evidence of bone marrow suppression was observed [20,21]. Our patient did not experience any of the mentioned side effects after 10 months of MMF treatment.
Although the second-line therapy with MMF seems promising, the response rates varied according to reason for stopping AZA treatment where patients with AZA intolerance had higher response rates to MMF than those who had shown insufficient response to AZA (43% vs. 25%) [18,20]. In our case, pancytopenia and alopecia occurred after 3 months of standard treatment (corticosteroid and AZA). AZA was discontinued and the patient was stable with prednisolone 5mg daily alone. Unfortunately after 27 months of corticosteroid monotherapy, hepatitis flare recurred and was stabilized with addition of AZA but again the same side effect, pancytopenia and alopecia developed. Like other studies, alternative treatment with MMF was considered as a good option and we expect better treatment response for our patient as she had shown sufficient response to AZA. However, further study based on larger number of patients is required.
Acknowledgements
This study was supported by a fund of the HBV cohort study [4800-4845-300-260, 2015-ER5101-00] from the Korea Centers for Disease Control and Prevention.
Notes
Conflicts of Interest: The authors have no conflicts to disclose.
Abbreviations
AIH
autoimmune hepatitis
ALP
alkaline phosphatase
ALT
alanine aminotransferase
ANA
antinuclear antibody
Anti-LKM
antibody anti-Liver Kidney Microsomal antibody
AST
aspartate aminotransferase
AZA
azathioprine
HAV Ab IgM
hepatitis A antibody immunoglobulin M
HBsAg
hepatitis B surface antigen
HCVAb
hepatitis C antibody
IgG
Immunoglobulin G
MMF
mycophenolate mofetil
PT INR
prothrombin time internal normalisation ratio
SMA
anti-smooth muscle antibody
TPMT
Thiopurine S-methyltransferase
6-MP
6-mercaptopurine
6-TG
6-thioguanine