What we need to know when performing and interpreting US elastography
Article information
Abstract
According to the increasing need for accurate staging of hepatic fibrosis, the ultrasound (US) elastography techniques have evolved significantly over the past two decades. Currently, US elastography is increasingly used in clinical practice. Previously published studies have demonstrated the excellent diagnostic performance of US elastography for the detection and staging of liver fibrosis. Although US elastography may seem easy to perform and interpret, there are many technical and clinical factors which can affect the results of US elastography. Therefore, clinicians who are involved with US elastography should be aware of these factors. The purpose of this article is to present a brief overview of US techniques with the relevant technology, the clinical indications, diagnostic performance, and technical and biological factors which should be considered in order to avoid misinterpretation of US elastography results.
INTRODUCTION
Hepatic fibrosis correlates with the severity of chronic liver damage. In the era of antiviral and antifibrotic therapy, noninvasive detection and quantification of hepatic fibrosis is becoming more and more important [1]. Although liver biopsy is the current gold standard for assessing hepatic fibrosis, the problems of liver biopsy are its high rates of sampling errors, invasiveness, difficulty to repeat, and observer variability. Therefore, liver biopsy is not an ideal tool for screening, longitudinal monitoring, and assessing the therapeutic response. The ideal test for the staging of hepatic fibrosis should be noninvasive, simple, readily available, not expensive, reproducible, and accurate. Given these conditions, US elastography have many advantages in becoming the ideal test for quantifying hepatic fibrosis.
US elastography techniques have evolved significantly over the past two decades. US elastography is not only an active area of research, but has also been implemented in clinical practice. According to its rapid development and adaptation from engineering and many vendors, variable terminology and parameters have been used, which can be very confusing. Recently, the European Federation of Societies for Ultrasound in Medicine and Biology (EFSUMB) [2,3], the World Federation for Ultrasound in Medicine and Biology and Biology (WFUMB) [4,5], and the Society of Radiologists in Ultrasound [6] have proposed guidelines and recommendations for US elastography. Beginning in 2012, the Quantitative Imaging Biomarkers Alliance (QIBA) of the Radiological Society of North America also organized the Ultrasound Shear Wave Speed (SWS) Biomarker Committee (http://qibawiki.rsna.org/index.php/Ultrasound_SWS_Biomarker_Ctte) as part of its continuing efforts to improve the value and practicality of the quantitative values obtained from US elastography. European experts also suggested an extension of the Standards for Reporting of Diagnostic Accuracy Studies (STARD) for reporting diagnostic accuracy studies on liver fibrosis tests, i.e. the Liver FibroSTARD standards [7]. In this review, the terms defined by the WFUMB are mainly used in order to facilitate the uniformity of the description (Table 1).
Although US elastography may seem easy to perform and interpret, it’s not a magic calculator. As there are many technical and clinical factors which can affect the results of US elastography, the interpretation of US results without appropriate knowledge might lead to misinterpretation. The purpose of this article is to present a brief overview of US techniques with their relevant technology, clinical indications, diagnostic performance, and technical and biological factors which should be considered in order to avoid misinterpretation.
OVERVIEW OF US ELASTOGRAPHY TECHNIQUES
US elastography techniques can be categorized into the stain imaging and the shear wave imaging according to the physical quantity each technique measures (Table 2). As strain imaging generally does not provide the absolute values of liver stiffness, it will not be further discussed in this review.
For the shear wave imaging, the physical quantity which is measured is the velocity of shear waves, and/or Young’s modulus (kPa) is converted from shear wave speed based on assumptions of constant density, homogeneity, isotropy, and static deformation using the equation for Young’s elastic modulus (E=3ρv2, where ρ is the shear wave velocity (m/sec) and ρ is the density of tissue, assumed to be constant) [8,9]. As we can see in this formula, the velocity of the shear wave increases with the stiffness of the liver parenchyma.
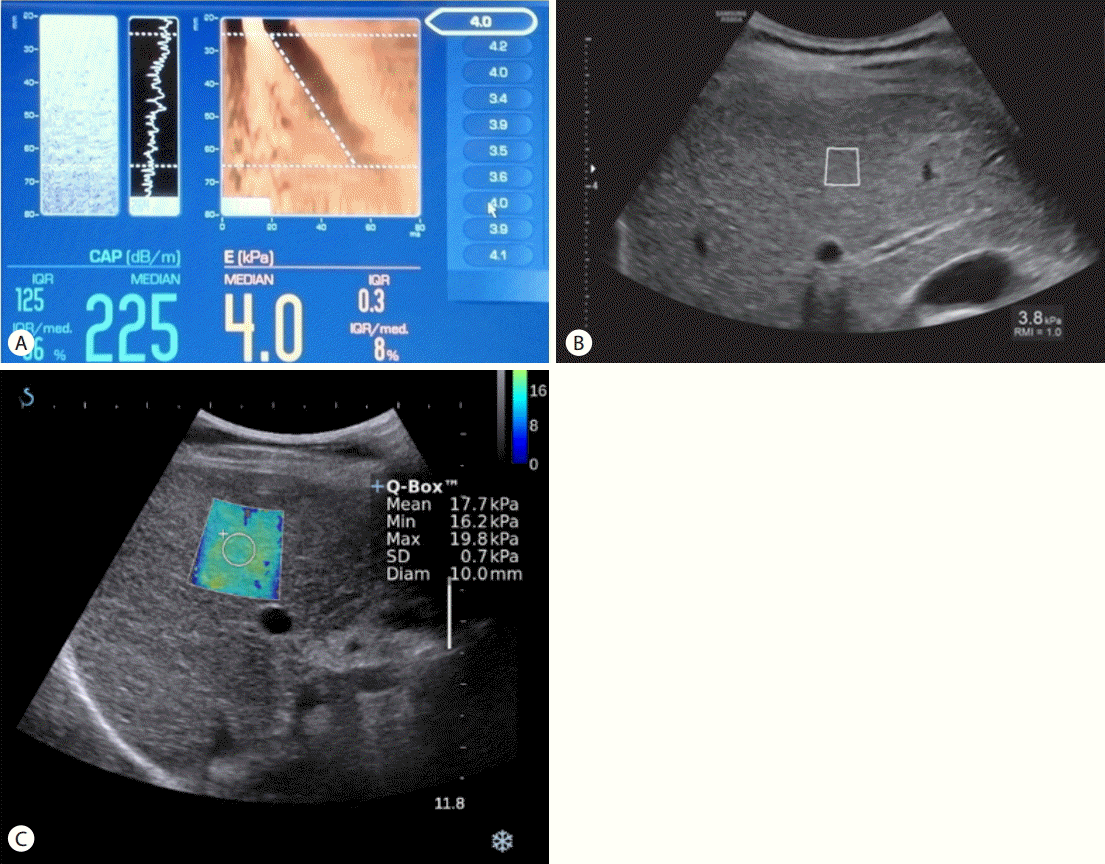
There are three methods for the shear wave imaging, i.e. transient elastography (TE); point shear wave speed measurement; and shear wave speed imaging. In order to generate shear waves, shear wave imaging can use a controlled, vibrating, external vibration in TE (Fibroscan, Echogen) or an acoustic radiation force impulse (ARFI) in point shear wave speed measurement (Virtual Touch Quantification, Siemens Healthcare; ElastPQ, Philips; Smart-Shear-wave elastography, Samsung medison, Seoul, Korea) and shear wave speed imaging (ShearWave Elastography, Aixplorer Super-Sonic Imagine; Virtual Touch Image Quantification, Siemens Healthcare; Shear Wave elastography, GE Healthcare; Shear Wave Elastography, Toshiba Medical Systems, Tochigi Otawara, Japan) (Table 2) (Fig. 1). While TE is not integrated into gray scale US, the method using ARFI excitation is integrated with gray scale US with conventional US probes (Fig. 1). The point shear wave speed measurement provides liver stiffness values of only a region of interest, while shear wave speed imaging shows real time imaging with a simultaneous display of gray scale US and a color elastogram map in which a quantitative measurement is possible (Fig. 1).
CLINICAL INDICATIONS FOR US ELASTOGRAPHY
Noninvasive assessment of liver fibrosis
The main clinical indication for US elastography is the noninvasive assessment of liver fibrosis in patients with chronic liver disease. The histologic fibrosis grade is well correlated with the liver stiffness values measured on US elastography [10,11]. The diagnostic performance of US elastography will be discussed in the following text.
Longitudinal monitoring of the treatment response and disease progression
Owing to its noninvasiveness, liver stiffness measured by US elastography is useful for assessing the longitudinal monitoring of the treatment response and disease progression [12-16]. In a study of 416 chronic hepatitis C patients, the sustained virological response with antiviral therapy was associated with a significant decline of liver stiffness seen on TE [13].
Portal hypertension and prediction of complications of cirrhosis
The standard method for evaluating portal hypertension is the measurement of the hepatic venous pressure gradient (HVPG) which, however, is an invasive technique requiring hepatic venous catheterization. US elastography can be a noninvasive alternative used to estimate the severity of portal hypertension and to predict complications associated with liver cirrhosis. In previously published studies, liver stiffness measurement by TE was positively correlated with HVPG [17,18]. The AUROC for the prediction of HVPG ≥10 mmHg or ≥12 mmHg was 0.99 and 0.92, respectively, with liver stiffness cutoff values of 13.6 kPa (sensitivity: 97%) and 17.6 kPa (sensitivity: 94%) in patients with hepatitis C [18]. In addition, liver stiffness measured by TE was correlated with the presence of esophageal varices in patients with hepatitis C virus (HCV)-related cirrhosis [17,19]. Therefore, US elastography can be helpful to stratify the risk for the development of varices and can be indicative of decompensated cirrhosis in cirrhotic patients.
DIAGNOSTIC PERFORMANCE AND CUT-OFF VALUES OF US ELASTOGRAPHY
TE
TE is the most extensively validated method among the US elastography techniques as it is a prototype of US elastography. Numerous studies and meta-analyses have demonstrated that liver stiffness values are strongly correlated with the histologic stage of fibrosis in patients with chronic hepatitis B virus (HBV) [20-26]. According to a recent meta-analysis of the performance TE in patients with chronic hepatitis B, the estimated cutoff for F2 (significant fibrosis) was 7.0 kPa (sensitivity, 78%; specificity, 80%), F3 was 8.8 kPa (sensitivity, 74.0%; specificity, 63.8%), and F4 was 11.7 kPa (sensitivity, 84.6%; specificity, 81.5%) [27] (Table 3). The diagnostic performance of TE is similar in patients with chronic HBV and HCV [28], although cut-off values were different according to the underlying causes of liver cirrhosis. Based on measurements in healthy volunteers from the general population, the mean liver stiffness value has been estimated to be 5.5±1.6 kPa [29]. The reproducibility of TE was excellent for both intraobserver and interobserver agreement with intraclass correlation coefficients (ICC) generally above 0.9 [30,31].
Point shear wave speed measurement
Among the US elastography techniques using point shear wave speed measurement, Virtual Touch Quantification (Siemens Healthcare) has been well validated. In a recent meta-analysis comprised of eight studies including 518 patients, the mean diagnostic accuracy of Virtual Touch Quantification expressed as AUROC was 0.87 (95% CI, 0.83-0.92) for the diagnosis of signifiant fibrosis (F ≥2), 0.91 (95% CI, 0.86-0.96) for the diagnosis of severe fibrosis (F ≥3), and 0.93 (95% CI, 0.89-0.97) for the diagnosis of cirrhosis [32]. The mean value of shear wave velocity using Virtual Touch Quantification in normal liver was lower than 1.3 m/sec [33-35]. Virtual Touch Quantification reproducibility was excellent in both intraobserver and interobserver agreement with the ICC generally above 0.8 (0.84-0.87) [33,36,37]. According to these results, Virtual Touch Quantification has been demonstrated to be a successful diagnostic tool for staging hepatic fibrosis.
Compared to the results from Virtual Touch Quantification (Siemens Healthcare), those from the other point shear wave speed measurement techniques are limited, as they have been recently introduced [38,39].
Shear wave speed imaging
ShearWave Elastography (Aixplorer supersonic imagine, France) is the most validated method among the shear wave speed imaging techniques. A previously published report demonstrated that the cut-off values of SWE were 7.1 kPa for significant fibrosis (F≥2), 8.7 kPa for advanced fibrosis (F≥3), and 10.4 kPa for cirrhosis (F=4) in chronic hepatitis C patients with the AUROC of 0.92 for F≥2, 0.98 for F≥3, and 0.98 for F≥4 [40]. The normal liver stiffness value ranged from 2.6-6.2 kPa assessed using SWE [41]. The reproducibility of ShearWave Elastography is excellent with an ICC above 0.90 [41-43].
Method of measuring US elastography
Patients should fast for 4-6 hours before US elastography, as food intake can increase the liver stiffness value. Right lobe measurements using the intercostal approach which is unaffected by the probe compression against the liver parenchyma, are preferred. Elastography measurements are acquired during breath-holding in order to minimize liver motion. Breath-hold at the end of expiration is preferable in order to determine the persistent liver position between acquisitions and to avoid overestimation of the liver stiffness. Having a clear sonic window is important in the sense that adequate shear wave generated by an ARFI push pulse is required for accurate measurement. The measurement depth can be a confounding factor for assessing liver stiffness [44]. According to the Society of Radiologists in Ultrasound, the ARFI pulse has a sweet spot at a 4-5 cm depth with most US equipment [6].
For TE, successful measurements are validated using the following criteria: the number of valid shots ≥10; the ratio of valid shots to the total number of shots ≥60%; and an interquartile range (IQR, reflecting the variability of measurements) less than 30% of the median liver stiffness measurement (LSM) value (IQR/LSM ≤ 30%). However, for the other US elastography techniques, the minimum number of measurements and the reliability criteria have not yet been well defined. According to the WFUMB recommendations [4,5], a median value of 5-10 measurements can be considered as a representative value in point shear wave speed measurement and the mean value of four measurements in shear wave speed imaging.
CONFOUNDING FACTORS AFFECTING THE PERFORMANCE OF US ELASOGRAPHY
Technical factors
Different techniques and vendors
Although liver stiffness measured by US elastography may appear as a unique and fixed physical property of the liver, in fact, the liver stiffness values measured by US elastography are a function of the frequency of shear waves [45,46]. As different US elastography techniques and vendors use different shear wave frequencies, the liver stiffness values from different vendors are consequently not exchangeable.
Measurement location
Liver stiffness is recommended to be measured in the right lobe of the liver as it is not altered by the effect of the probe compression against the liver. Liver stiffness measured in the left lobe is higher than that in the right lobe [35,37,47-49]. The region of interest (ROIs) should be placed in a homogenous area without vessels and artifacts. This principle should be applied to the path of push pulses as well as to ROIs, as the area between the push pulse and the ROI should be homogenous in order to obtain the good shear wave propagation.
The depth of measurement can be a confounding factor for assessing liver stiffness. A previously published study reported that liver stiffness had a decreasing tendency according to the increased depth of measurement in the same phantom [44]. Significant differences were found in all of the different types of US elastography according to the depth of measurement in each vendor’s machine and each transducer in the same phantom [44]. Interaction between liver stiffness and the fibrosis stage is more accurate for measurements at 1-3 cm below the liver capsule than for superficial measurements between 0 and 1 cm below the liver capsule [50]. There can also be artifacts at the area just beneath the liver capsule. Therefore, the depth of measurement should be 2-5 cm below the liver capsule. According to the Society of Radiologists in Ultrasound, the ARFI pulse has a sweet spot at 4-5 cm depth with most equipment [6].
Patient factors
Inflammatory activity
US elastography is unreliable for detecting liver fibrosis in patients with acute hepatitis [51-53]. As inflammatory changes, including edema, can increase the elasticity and viscosity, inflammatory activity has a major impact on liver stiffness. Therefore, acute liver damage can lead to overestimation of the fibrosis stage. Some authors have recommended using different cutoff values of liver stiffness with respect to the alanine aminotransferase (ALT) level [21]. A higher liver stiffness cutoff is required in order to diagnose different degrees of liver fibrosis in patients with elevated ALT levels.
Biliary obstruction
Obstructive cholestasis secondary to benign and malignant etiologies can reversibly increase liver stiffness [54-58], as it causes secondary fibrotic or inflammatory changes.
Liver steatosis
The influence of steatosis on liver stiffness values measured by US elastography is currently controversial, as conflicting results exist. Some report have suggested that US elastography was less accurate for detecting severe fibrosis in NAFLD patients [59,60]. However, other studies have reported that liver stiffness was not affected by the presence of hepatic steatosis [61,62].
Hepatic venous congestion
Hepatic venous congestion can influence US elastography performance [63,64]. Deformation of hepatic vessels in sinusoidal diseases or vascular disease can increase the liver stiffness. Sinusoidal obstruction syndrome can reversibly increase liver stiffness measurements [65,66]. In order to avoid overestimation of the liver stiffness, US elastography should be interpreted with caution in patients with hepatic congestion and congestive heart failure.
Liver infiltration
We must keep in mind that liver stiffness measurement is not specific to liver fibrosis. Diffuse infiltrative liver disease, such as amyloidosis, can also increase the liver stiffness [67].
Deep inspiration
Deep inspiration has been shown to increase stiffness measurements compared with a resting expiratory position [35].
Food intake
Food intake was seen to significantly increase the liver stiffness measurements in patients with chronic liver disease as well as in healthy controls [68-71], as it was associated with an increase in splanchnic and hepatic blood circulation.
Body habitus of patients
Owing to the fixed depth of measurement in TE, TE is known to be of limited use in patients with ascites, thick subcutaneous fat, narrow intercostal space, and severe obesity [10,31,72]. A TE XL probe has been introduced and validated for obese patients, and the use of an XL probe reduces the failure rate in obese patients but results in a high rate of unreliable results (approximately 25%) [5]. Body habitus, i.e. narrow intercostal space and severe obesity, can also affect other US elastography methods [73,74].
CONCLUSION
US elastography is useful in the assessment of the severity of hepatic fibrosis, longitudinal monitoring of the treatment response and disease progression, estimation of portal hypertension, and predicting the complications associated with liver cirrhosis. In order to maintain the successful and valuable performance of US elastography demonstrated in previously published studies, understanding its operating principles and technical details is crucial. It is necessary to be cautious when interpreting the parameters of US elastography measured under different conditions as there are many factors affecting the results of US elastography.
Notes
Conflicts of Interest: The authors have no conflicts to disclose.
Abbreviations
ALT
alanine aminotransferase
ARFI
acoustic radiation force impulse
EFSUMB
European Federation of Societies for Ultrasound in Medicine and Biology
HBV
hepatitis B virus
HCV
hepatitis C virus
HVPG
hepatic venous pressure gradient
ICC
intraclass correlation coefficient
IQR
interquartile range
LSM
liver stiffness measurement
NAFLD
nonalcoholic fatty liver disease
PSWSM
point shear wave speed measurement
QIBA
Quantitative Imaging Biomarkers Alliance
STARD
Standards for Reporting of Diagnostic Accuracy Studies
SWS
Shear Wave Speed
TE
transient elastography
US
ultrasound
WFUMB
World Federation for Ultrasound in Medicine and Biology and Biology