The management of post-transplantation recurrence of hepatocellular carcinoma
Article information
Abstract
The annual incidence of hepatocellular carcinoma (HCC) continues to rise. Over the last two decades, liver transplantation (LT) has become the preferable treatment of HCC, when feasible and strict selection criteria are met. With the rise in HCC-related LT, compounded by downstaging techniques and expansion of transplant selection criteria, a parallel increase in number of post-transplantation HCC recurrence is expected. Additionally, in the context of an immunosuppressed transplant host, recurrences may behave aggressively and more challenging to manage, resulting in poor prognosis. Despite this, no consensus or best practice guidelines for post-transplantation cancer surveillance and recurrence management for HCC currently exist. Studies with adequate population sizes and high-level evidence are lacking, and the role of systemic and locoregional therapies for graft and extrahepatic recurrences remains under debate. This review seeks to summarize the existing literature on post-transplant HCC surveillance and recurrence management. It highlights the value of early tumour detection, re-evaluating the immunosuppression regimen, and staging to differentiate disseminated recurrence from intrahepatic or extrahepatic oligo-recurrence. This ultimately guides decision-making and maximizes treatment effect. Treatment recommendations specific to recurrence type are provided based on currently available locoregional and systemic therapies.
INTRODUCTION
Primary liver cancer is the third most common cause of cancer-related deaths worldwide [1], with hepatocellular carcinoma (HCC) being a significant contributor as the most common primary liver cancer type [2,3]. The annual incidence of HCC is rising, with an overall three-fold increase over the last three decades [2,4]. Liver transplantation (LT) has emerged as the preferred treatment for early-stage HCC due to the dual benefit of removing both the primary tumour and the underlying tumourigenic cirrhotic environment, which results in favorable disease-free survival [5-8]. HCC continues to grow as an indication for LT, currently accounting for at least 22% of all LT indications in the USA [5], and 32.4% of all LT in Canada [9].
Despite stringent institutional transplant eligibility criteria, 6–18% of transplanted patients develop HCC recurrence, and risk estimation models remain only partially predictive [4, 6-8,10]. Milan criteria is the gold standard for selection of HCC patients for liver transplants to minimize HCC recurrence rates [11,12]. Recently, more centres are transplanting patients beyond the Milan criteria based on various extended transplant criteria, or are downstaging via locoregional therapies from beyond to within the Milan criteria [6,13,14]. The increasing utilization of expanded criteria and downstaging further increases the number of HCC-related LT [15]. Consequently, this increases the overall number of patients who develop recurrence.
HCC recurrence is an essential prognosticator for post-transplantation survival [16], leaving a median survival of 10–13 months following recurrence [17-19]. Currently, the clinical management of HCC recurrence is challenging [20] as standardized protocols for post-LT surveillance and consensus treatment guidelines are lacking [21]. Through this literature review, we aim to provide a summary of the existing literature on management of post-transplant HCC recurrence, including surveillance strategies for detecting recurrence, re-evaluating the post-transplant immunosuppression regimen, and staging to distinguish disseminated and oligo-recurrence, to cater management practices specific to recurrence type.
POST-TRANSPLANT SURVEILLANCE FOR HCC RECURRENCE
Emerging data and expert consensus supports post-transplant HCC surveillance, as increased cumulative surveillance exposure, early diagnosis, and aggressive treatment have been demonstrated to improve survival outcomes [16,22,23]. The American Association for the Study of Liver Diseases (AASLD) guidelines recommend the use of the externally validated Risk Estimation of Tumor Recurrence After Transplant (RETREAT) prognostication score [24] to determine a patient’s 5-year recurrence risk and guide optimal screening intervals [25]. Multiple other prognostic risk scoring systems for HCC recurrence post-LT have been developed on a per-protocol analysis [20,26]. The most prominent prognostic risk scoring systems are outlined in Table 1. These scoring systems can provide guidance for post-operative surveillance strategies. However, despite identifying risk factors and prognostic models for HCC recurrence, there is limited direct application into clinical practice [27-29]. Currently, consensus surveillance protocols for the frequency and duration of surveillance for HCC recurrence do not exist [27-29].
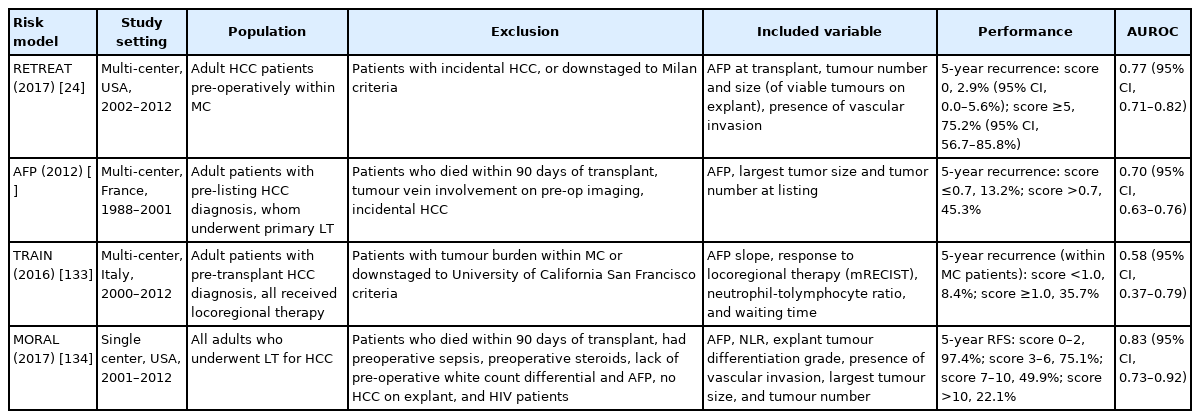
A summary of common existing prognostic risk scoring systems for HCC recurrence following liver transplantation, which can provide guidance for post-operative surveillance strategies
The most frequent site of HCC recurrence is extrahepatic alone (50–60%), commonly lungs and bone, followed by combined extrahepatic and intrahepatic (30–40%), and intrahepatic only (15–40%) [30,31]. Multiphase computed tomography (CT) scans can play a critical role in monitoring HCC recurrence [32]. Currently, the American guidelines recommend post-transplant surveillance for HCC recurrence with chest and abdominal CT scans, though optimal timing and duration is uncertain [25]. Additionally, elevated serum biomarkers, including alpha-fetoprotein (AFP) levels, have been shown to correlate with HCC recurrence, independent of the timing or location of the recurrence [33]. AFP levels have been validated for use as a predictor for HCC recurrence [34], and AFP ≥100 U/L at the time of recurrence have been correlated with worse overall survival (OS) (hazard ratio, 1.689; 95% confidence interval, 1.059–2.695; P=0.028) [35]. There may also be future potential in analysis of molecular biomarkers including DNA alterations, aberrant gene expression, microRNA, and circulating tumour cells, towards HCC risk stratification and outcomes prediction [36].
HCC recurrence occurs most frequently (60%) in the first 2 years post-transplant [12,33,37], and this early recurrence is predictive of worse prognosis, often with the increased disease burden and extrahepatic metastases [17,19,38], One recent study by Kim et al. [35], demonstrated median timing of recurrence for single intrahepatic (20.6 months; interquartile range [IQR], 9.8–32.1), multiple intrahepatic (9.6 months; IQR, 5.2–14.4), single extrahepatic (11.1 months; IQR, 4.6–19.0), and single lung recurrence (21.4 months; IQR, 10.4–41.1). Late recurrence (beyond 2 years) have also been reported and may have more favorable tumour biology, with better response to locoregional treatments [33,39]. Given the greater proportion of early recurrences and the observed worse prognosis, current data supports intense surveillance for the first 2 years post-transplant, to identify early recurrences [29,33,37].
Aggarwal et al. [40] performed a national survey of post-transplant HCC surveillance patterns amongst 48 transplant centres across the USA, and found that 96% of centres had an existing surveillance protocol. Most centres (74%) included cross-sectional imaging of both chest and abdomen, whereas 21% did abdominal only, and 3% additionally incorporated a bone scan [40]. Sixty-five percent of centers incorporated AFP levels within their surveillance protocol. Forty-eight percent of centers reported 5-year duration for surveillance, while 18% discontinued surveillance within the initial 2 years [40]. The most commonly implemented surveillance strategy involved imaging every 3–4 months for the first year, every 6 months for the second year, and every 6–12 months for the following 3 years. In those deemed to have higher recurrence risk, surveillance was more frequent with chest and abdominal CT imaging every 3–6 months for the initial 5 years [40]. Studies have demonstrated no difference in performing CT scans every 3 versus every 6 months for detecting HCC recurrence [22,41].
One study, which combined CT scans and AFP levels for HCC screening every 6 months, showed that surveillance over 2 years incurred high costs compared to life years gained [42]. Given the low rate of HCC recurrence, the authors inferred that surveillance yields the greatest cost-benefit in the first 2 years following LT. The most critical variable noted was the survival benefit gained from finding resectable recurrence [42]. However, other studies from high volume LT centres support the significance of late-occurring recurrence, and recommend surveillance with cross-sectional imaging and AFP every 3–6 months post-transplant, for a minimum of 5 years [33,43,44]. Additionally, predicting an individual’s risk for post-transplant HCC recurrence through risk stratification prognostication scores [24,25] may be necessary in guiding personalized surveillance strategies [33]. Currently, few transplant centers stray away from institution-specific routine screening practices towards individualized surveillance strategies, based on patient risk stratification for post-transplant HCC recurrence [40]. Overall, the knowledge gap and lack of established surveillance guidelines have led to significant heterogeneity in surveillance patterns across institutions, with ongoing debate regarding the appropriate surveillance methods, frequency, and duration [33,40].
THE INTERPLAY OF IMMUNOSUPPRESSION AND HCC RECURRENCE
The role of immunosuppression is an important consideration in post-transplant HCC recurrence. The adaptive immune system provides the body’s standard defence against tumour cells. Additionally, in cases of recurrence concomitant immunity exists, whereby initial encounter of the primary tumour induces growth inhibition of further secondary tumours or metastases though anti-tumour immune response and immunosuppressive cellular mechanisms [45,46]. In the post-transplant state, both standard and concomitant immunity are suppressed, which may account for the more aggressive and faster progression of HCC recurrence posttransplant compared to post-resection [47]. However, post-transplant immunosuppression is critical in preventing graft rejection and dysfunction. This necessitates revisiting the immunosuppression strategy on diagnosis of post-transplant recurrence. The goal is to maximize the benefits of an active immune response for minimizing tumour progression, without compromising graft function.
The current mainstay for immunosuppression following LT include calcineurin inhibitors (CNI), such as tacrolimus and cyclosporins [48,49]. However, CNIs can create a permissive environment for tumour growth, with increased risk for HCC recurrence in a dose-dependent manner [48-52]. A role for aberrant mammalian target of rapamycin (mTOR) signalling pathway, involved in cellular growth and proliferation, has also been shown to play a role in the pathogenesis of HCC [53-55]. Various mTOR inhibitors (mTORi) have been developed as immunosuppressive agents including sirolimus, and everolimus, which have shown to also have anti-tumour growth effects through suppression of cellular proliferation and angiogenesis [56,57]. Multiple cohort studies demonstrated decreased risk for post-transplant HCC recurrence and longer post-recurrence survival using mTORi compared to CNIs [58-60]. One retrospective study by Yang et al. [60], 2020 showed that compared to tacrolimus-based immunosuppression, sirolimus improved OS at 1 and 2 years following post-transplant HCC recurrence (P=0.035). Contrarily, a large international multicentre randomized control trial (the SiLVER trial), investigating sirolimus-based versus mTORi-free immunosuppression in LT for HCC, demonstrated no significant difference in overall or recurrence-free survival with mTORi [61]. However, the benefits of mTORi may be dependent on timing of recurrence. Kim et al. [35], demonstrates that everolimus initiation within 3 months following recurrence demonstrated improved OS in patients with a recurrence free duration >9 months (P=0.020), and no difference in those with recurrence within 9 months (P=0.149).
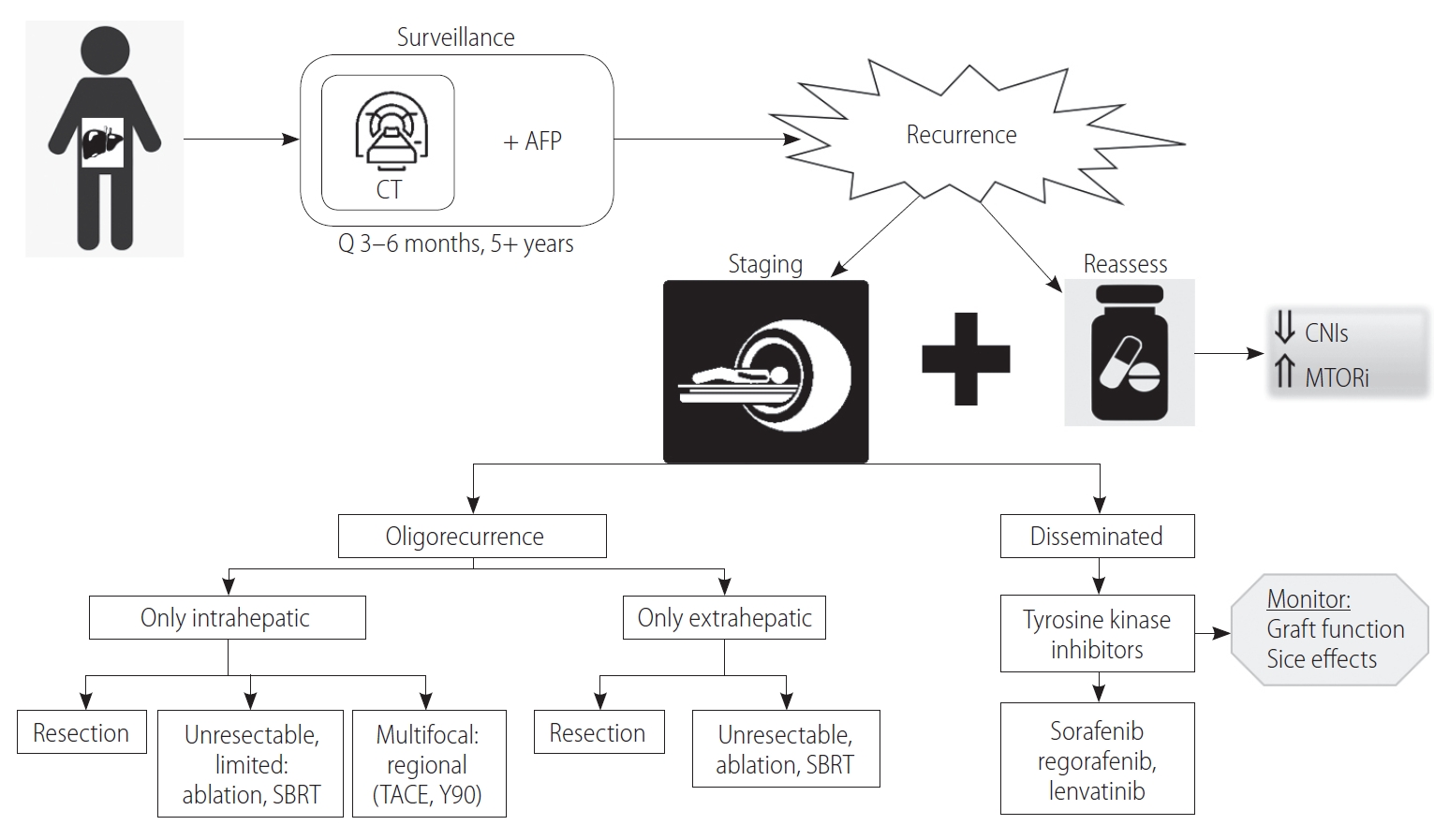
With HCC recurrence post-transplant, the current recommendation is to reconsider the immunosuppression regimen, and taper immunosuppression to the lowest effective dose for protection against graft rejection [6]. Specifically, this can be done by combining or completely switching to an mTORi such as sirolimus, and decreasing the CNI dosage [6,47]. Berenguer et al. [6] further recommended keeping CNI trough levels below certain targets: <10 ng/mL for tacrolimus, and <300 ng/mL for cyclosporin. However, the caveat is that close monitor of graft function and toxicity is required, and immunosuppression strategies should be individualized. Additionally, the administration of mTORi may have most survival benefits in patients who develop HCC recurrence beyond 9 months [35], though further studies are required.
STAGING OF DETECTED POST-LT HCC RECURRENCE
Historically, post-transplant recurrence was defined as a distant metastasis and considered terminal in prognosis, with palliative intent management [47]. Nowadays, recurrence is classified as either oligo-recurrence (further classified as intrahepatic and/or extrahepatic recurrence) or disseminated recurrence. This notion of differentiating limited and disseminated disease has led to a paradigm shift in management [47]. Studies have demonstrated that in those post-transplant HCC recurrence, eligibility for treatment with curative intent was a key predictor of survival, compared to treatment with palliative intent, or best supportive care [17,62]. Although many studies support aggressive treatment of post-transplant HCC recurrence by combining surgical and non-surgical therapies towards improved long-term survival [10,17,22,60,63], there is considerable debate on specific management practices, as standardized protocols do not exist.
Complete staging is essential for adequate decision-making in the management of post-transplant HCC recurrence. The distinction between limited (an oligo-recurrence) and disseminated recurrence further guides treatment eligibility for locoregional or systemic therapies [47]. Comprehensive staging post-transplant for detecting HCC recurrence is generally performed by combining cross-sectional CT imaging with skeletal examination by a bone scan [47]. Once completely staged, a systemic approach for management based on the type of recurrences at hand, i.e., disseminated vs. oligo-recurrence, and intrahepatic vs. extrahepatic vs. combined, should be followed to guide appropriate decision-making.
MANAGEMENT OPTIONS IN POST-TRANSPLANT HCC RECURRENCE
Intrahepatic oligo-recurrence
For oligo-recurrent disease, especially when confined to the liver, the selection of any given therapy is individualized, and depends on the tumour location, degree of disease burden, technical operative considerations, functional residual liver volume, and the patient’s functional status [27]. The various modalities available for curative intent include graft surgical resection and ablation. Other therapies are with palliative intent, and include external beam radiation, regional therapies: trans-arterial chemoembolization (TACE), and intra-arterial Yttrium-90 radioembolization (Y90), and combination therapy with systemic treatments: molecular-targeted therapies, and immunotherapy.
Surgical resection
Around 15–20% of post-transplant HCC recurrence is localized disease [19,23,64]. Graft tumour resection has been shown to have survival benefits in intrahepatic oligorecurrence [23,64], with prolonged long-term 3- and 5-year survival [65-70]. Cohort studies have demonstrated a median survival of 20–27 months in those who received surgical therapy, compared to 9–10 months in those receiving non-surgical therapy (other locoregional and systemic therapies), and 2.4–3.7 months in those that received best supportive care (no cancer treatment) [10,60]. These studies demonstrate survival benefits of aggressive surgical intervention in well-selected patients with post-transplant HCC recurrence [10,60,65-70]. However, these studies are subject to a high degree of selection bias. Surgical resection was more likely to be performed in patients with better functional status, fewer recurrence nodules, and those that developed late HCC recurrence, which are factors associated with an improved prognosis [60].
Surgical management in the post-transplant setting may involve extensive hilar adhesions, and also requires that the remnant liver is tumour-free with adequate functional residual volume [71-73]. These pose several operative challenges. Additionally, surgical resection contributes to significant overall post-operative morbidity (60–80%) [65-68], with patients being at higher risk for infective complications in the setting of immunosuppression. Prospective randomized trials are currently lacking, and there is a need for strong quality evidence on surgical resection in the management of oligo-recurrences.
Ablation
Surgical resection is considered the most favourable for curative treatment of localized HCC recurrence. However, when surgery is contraindicated or is technically not feasible, ablation technologies including radiofrequency ablation (RFA), microwave ablation, and irreversible electroporation can alternatively be used. Ablation is ideal for tumours located away from adjacent organs and major vascular structures due to the heat sink effect [47]. It is more preferable to resection for deep parenchymal tumours, for which resection would require a major hepatectomy [47]. Ablation is also less morbid than post-transplant liver resection, as it is minimally invasive, which avoids the need to perform a major laparotomy in an immunocompromised patient [74-76]. Tumour size (<3 cm), tumour number, and the presence of limited extrahepatic metastasis are the key predictors of treatment efficacy and prognostic factors of overall morbidity and mortality associated with ablation therapy [41,75].
One small, single centre retrospective study comparing eleven patients who received RFA to 15 patient with surgical resection for post-transplant HCC recurrence have demonstrated comparable long-term outcomes [47,67]. This study demonstrated similar 1-, 3-, and 5-year OS (92%, 51%, and 35% surgery vs. 87%, 51%, and 28% RFA) and 1-, 3-, and 5-year recurrence-free survival (83%, 16%, and 16% surgery vs. 76%, 22%, and 0% RFA) [47,67]. A case series of 11 patients whom underwent microwave ablation and were followed up to 33 months, had an average 17.3-month survival, with mild side effects overall [77]. However, 15.8% of cases had local tumour progress [77]. All of these studies were limited in sample size to draw significant conclusions, but demonstrate the promising potential of various ablation techniques for post-transplant HCC recurrence, with limited complication rates [62,77]. This technique should be used in cases of small, single liver-only recurrences that are unresectable.
Radiation
Another option in the treatment of post-transplant HCC recurrence is stereotactic body radiation therapy (SBRT). SBRT typically involves image-guided focal radiation to a tumour, with benefits of minimal collateral damage [78]. Additionally, SBRT has been shown to upregulate the tumour immunity response through stimulation of tumour-specific cytotoxic T cells [79,80]. Unlike RFA, SBRT can be effective for large tumour sizes [81], and has been used in several prospective studies for primary HCC in the non-transplant setting [81-83]. It has been shown to be effective for tumours ranging from 2–7 cm in size, acquiring 80–95% local tumour control at 2 years post-treatment [81-83]. One meta-analysis demonstrates that SBRT has equivalent OS and better local control compared to RFA in the HCC non-transplant setting, when adjusted for Barcelona Clinic Liver Cancer staging factors of liver function, performance status, and tumour size [84]. This highlights SBRT as a promising potential treatment strategy for local tumour control. However, all these studies were in the non-transplant population, and caution should be taken when extrapolating to the posttransplant setting.
Au et al. [85], 2020 performed a retrospective study of six patients with intrahepatic post-transplant HCC recurrence treated with SBRT. The authors demonstrated that there was no local progression or mortality at 15.5 months (median follow up duration) [85]. However, six treated lesions had regional progression (67%) and two patients had distant spread to the lungs (22%) [85]. Currently, there are limited studies with very small population numbers, and further studies are needed in investigating the role of SBRT in post-transplant HCC recurrence [85].
Regional therapies
For post-transplantHCC with multifocal intrahepatic recurrence, regional treatments including TACE and Y90 can be offered [86-88]. One prospective study of unresectable intrahepatic post-transplant HCC recurrence demonstrated a survival benefit with TACE compared to systemic therapy alone [86]. TACE can be difficult to administer safely post-transplant due to dense hilar adhesions, variable vascular anatomy, and the need to negotiate the catheter through the arterial anastomosis [66]. There are concerns for associated hepatic artery damage, with stenosis or occlusion, which affects OS [89]. There is also a potential risk for graft failure with the administration of TACE in the post-transplant setting. However, one systematic review demonstrated that TACE for post-transplant HCC recurrence was well tolerated without major adverse consequences [27]. Furthermore, in patients with unresectable HCC who are ineligible for TACE, a recent systematic review and network meta-analysis with three randomized controlled trials demonstrated no significant difference in treatment with systemic targeted therapies (sorafenib, lenvatinib) compared to Y90 microspheres [90]. Though many studies are investigating TACE and Y90 in the bridging period before LT, there are currently limited studies investigating the role of regional therapies in the setting of post-transplant HCC recurrence.
Extrahepatic oligo-recurrence
With regards to extrahepatic HCC recurrence, the most common sites affected include: lungs (40–60%), bones (25–30%), adrenals (10%), lymph nodes (10%), and peritoneum (9%) [8,27,31]. Similar to intrahepatic oligo-recurrence, extrahepatic recurrence can also be treated with locoregional therapies including resection and radiation, and systemic therapies.
Surgical resection
Similar to other metastatic malignancies to the lung, locoregional therapies, including pulmonary metastasectomy, can be used towards the goal of curative intent in HCC-related pulmonary metastases, with prolonged survival outcomes [91,92]. Multiple retrospective cohort studies demonstrate that surgical resection is effective in post-transplant pulmonary recurrence with greater 2- and 5-year survival rates compared to no resection [47,92,93]. This held true even with multiple pulmonary sites for recurrence, as long as surgical resection left behind adequate lung function [47,92,93]. Similarly, there have been numerous case studies describing surgical resection for other locations of post-transplant extrahepatic HCC oligo-recurrences, including to the vertebra [94], adrenals [95,96], lymph node [97], and peritoneum [33]. Most of these case studies demonstrate favourable survival outcomes, however it is difficult to draw robust conclusions [33,95-97]. Though these cohort studies and case reports demonstrate efficacy of surgical resection in extrahepatic oligo-recurrence, the literature is limited by the low quality of the study design and the potential for confounding due to patient selection bias in these resection candidates. Consequently, further higher-evidence studies are required in assessing the role of surgical resection in extrahepatic HCC oligo-recurrence.
Radiation
In those not candidates for surgery, one alternative treatment strategy for local recurrence is SBRT. Though this is for non-curative, palliative intent, SBRT has demonstrated some efficacy for local recurrence control in lung and bone metastases [98]. However, the literature is limited to case reports. One case report of two individuals with HCC oligo-recurrence to lymph nodes describes the usages of SBRT given over three to five fractions for curative intent [99]. One individual had a complete response and remained cancer-free at 31 months follow-up. The second patient developed multifocal liver recurrence within 2 months of treatment, though had stable disease at the irradiated lymph node at 20 months [99].
Role of systemic therapy in oligo-recurrence
Systemic therapies such as molecular-targeted therapies or tyrosine-kinase inhibitors (TKIs) may be combined with locoregional treatments (surgical resection, ablation, SBRT), and regional therapies to treat intrahepatic and extrahepatic post-transplant HCC oligo-recurrence. One study by Yang et al. [60] demonstrated that the OS for oligo-recurrence was most favourable for patients receiving a combination of surgical resection followed by non-surgical therapy, including external beam radiotherapy for bone metastasis, TKIs, and sirolimus-based immunosuppression. The 1- and 2-year OS of combination therapy was 93.8% and 52.6% compared to 30.8% and 10.8% in patients receiving non-surgical therapy alone, P<0.001 [60]. The 2-year OS was an 80% in patients that had resection of their recurrent disease, followed by a sorafenib or lenvatinib therapy and sirolimus-based immunosuppression [60].
A systematic review including 61 studies (13 case reports, 41 case series, and seven retrospective comparative studies) demonstrated that the most favourable survival outcomes were achieved with surgical resection for the localized HCC recurrence [27]. Consequently, attempting surgical resection is recommended, where feasible [27]. Other locoregional and regional therapies, including ablation and TACE, can be safely performed in the case of unresectable disease or unfavourable surgical candidacy [27]. Locoregional therapies can also be combined effectively with systemic therapies such as sorafenib if patients can tolerate the side effects [27]. Combination of locoregional and systemic therapies are currently being investigated in the setting of primary HCC in the non-transplant population, through clinical trials [100]. This may also further influence the direction for future clinical trials on the role of systemic and combination therapy specific to the population of post-transplant HCC oligo-recurrences.
Disseminated HCC recurrence
Disseminated HCC recurrence involves the systemic spread of disease, which proposes a significant role for systemic therapies, including molecular-targeted therapies and immunotherapy. The goal is to prolong survival and treatment is for palliative intent, rather than pursuit of cure.
Systemic therapy
TKIs has been used in combination with other interventions for disseminated HCC recurrence [101,102]. Sorafenib was the earliest approved of the targeted therapies with clinically proven efficacy for unresectable HCC [103]. Multiple retrospective studies show that combining sorafenib with mTORi, and treating early in the disease course following post-transplant HCC recurrence, leads to disease stability or a complete or partial response [60,104-111]. These studies have demonstrated a survival benefit from systemic therapy ranging from 7.5–20 months, compared to best supportive care alone [60,104-111]. However, patients had significant drug toxicity, with poorly tolerated side effects and a 29% discontinuity rate [60,104-111]. Nonetheless, these studies were not performed with population matching, and can be confounded by patient selection bias.
One retrospective study performed at a large tertiary centre consisted of 41 patients with post-transplant HCC disseminated disease who received systemic therapy [38]. Seventy-nine percent of the patients received sorafenib and had a median OS of 14 months from recurrence, with 36% disease control [38]. The most common adverse events included hand-foot syndrome (34.7%), diarrhea (26.7%), and dose discontinuation or interruption (8.8%), with 47.1% of patients switching to a second-line systemic agent [38]. Two patients had biopsy-proven transplant rejection, that resolved by increasing the immunosuppressants [38]. Therefore, dosing of TKIs should be personalized and based on a risk-benefit patient discussion.
Many new systemic therapy drugs are also being approved for use as second-line for advanced or unresectable HCC in the non-transplant setting, following failure or tolerance of first-line sorafenib. These include regorafenib, which was approved in 2017 [112], and lenvatinib approved in 2018 through the international phase III trial [113]. The use of systemic therapies in post-transplant patients is complex due to both the immunosuppressive environment and risk for drug interactions between systemic therapies and immunosuppressants. Available data on these newer systemic drugs in the context of post-transplant HCC recurrence is limited, as systemic therapy trials have historically excluded transplant patients [6].
A recent multi-centered study by Iavarone et al. [114] showed clinical efficacy and safety of regorafenib use in post-transplant HCC recurrence for patients who developed tolerance and progressed on sorafenib treatment, demonstrating a median OS of 12.9 months following treatment initiation. Similarly, Yang et al. [60] showed a median OS of 19.5 months with lenvatinib, compared to those who discontinued or failed sorafenib, or those transitioned to regorafenib therapy (median OS of 12 months).
Immunotherapy
Another promising option for disseminated post-transplant HCC recurrence includes immunotherapy, which directs the host’s immune response towards the tumour, by prompting an immune reaction against the tumour antigens [115]. Various immunotherapies act as immune checkpoint inhibitors. One group of immunotherapy agents are programmed cell death protein (PD-1) blockers, including nivolumab, ipilimumab, and pembrolizumab, which have been validated in large phase two trials [115]. Nivolumab and pembrolizumab have also been recently approved for use in advanced, unresectable HCC in the non-transplant setting [116,117]. Furthermore, a recent global open-label phase three trial (IMbrave150 trial) demonstrated better overall and progression-free survival for PD-1 inhibitors atezolizumab plus bevacizumab compared to sorafenib alone [118]. PD-1 inhibitors have demonstrated overall favourable outcomes in patients that have failed sorafenib [116,119]. They also have a better side effect profile, and have demonstrated a 15–20% response rate, with 64% disease control for primary HCC [116,119].
Currently, there is a paucity of studies examining the role of immunotherapy in post-transplant HCC recurrence, with only a few existing case reports and series [120,121]. Consequently, it is challenging to draw conclusions on efficacy. There are concerns that immunotherapy regulates cell-mediated immunity, which can interfere with post-transplant immune tolerance and contribute to treatment-resistant allograft transplant rejection and injury [121-124]. One retrospective pilot study from Mayo Clinic (n=7) with post-transplant recurrence of HCC (n=5) or melanoma (n=2) demonstrated some preliminary efficacy of PD-1 inhibitors with complete response (one of four patients), though two of seven patients developed allograft rejection [121]. Further studies are needed to investigate the efficacy and safety of immunotherapy in the setting of post-transplant HCC recurrence. At this point immunotherapy in the transplant population can’t be recommended though future clinical trials are currently underway.
FUTURE DIRECTIONS
Clinical trials have previously excluded transplant patients [125], and much of the management strategy for post-transplant HCC recurrence is drawn from management principles of primary HCC in the non-transplant setting [29,37]. Consequently, there continues to be a paucity of literature and a lack of strong evidence. However, this review summarizes the currently available literature to help guide clinicians on post-transplant screening and treatment decision-making in the management of HCC recurrence. The envisioned ideal trajectory following LT for HCC should involve screening for recurrence, which should be personalized based on individual recurrence risk. Those with high recurrence risk should be screened every 3–6 months for the first 5 years. Once recurrence is detected, a patient’s immunosuppression regimen should be re-evaluated, and complete staging should be obtained to determine the presence of oligo-recurrence or disseminated disease. Depending on the type of recurrence, the patient’s functional status, and their wishes, either curative intent or palliative treatment can be pursued.
Many promising treatment options are on the horizon that have been approved for advanced HCC, but have yet to be studied in the post-transplant population with HCC recurrence. One challenge is managing this population in the context of the post-transplant immunosuppressed state. There is a sensitive balance between tumour recurrence and progression, with graft rejection or failure. Additionally, many studies have focused on risk factors and predictors for post-transplant HCC recurrence, but there is a significant lack of literature on management strategies and clinical application. Many studies have low-quality of evidence in the form of case reports, case series, or retrospective cohort design. These studies are often obscured by confounders and limited in the ability to draw any clinically-applicable conclusions. This highlights the need for future clinical trials investigating management of post-transplant HCC recurrence.
There are some ongoing clinical trials (Table 2) in the setting of post-transplantn HCC recurrence. Two clinical trials are assessing the safety and efficacy of PD-1 inhibitor use in those intolerant to or previously progressed on sorafenib [126,127]. Other trials are evaluating the role of TKIs, including a phase II study currently recruiting at Princess Margaret Hospital in Toronto, Canada, which is assessing the efficacy of cabozantinib [128], and a future phase III study evaluating relenvatinib [129]. Additionally, two trials in China are investigating post-transplant HCC recurrence in the context of hepatitis B virus liver etiology [130,131]. One is an open-label phase I study assessing a hepatitis B virus-specific T-cell receptor-redirected drug [130], while the other is a phase I/II multicentre study assessing a biologic, liocyx that lyses target liver cells [131].
CONCLUSION
As the number of LT for HCC increases, the number of patients experiencing post-transplant HCC recurrence will rise. Post-transplant HCC recurrence represents a significant clinical challenge and is associated with poor prognosis. Currently, there is no consensus best practice guidelines for HCC surveillance and recurrence management, partly due to limited existing high-level evidence. This review summarizes the available literature to inform and guide clinicians in managing post-transplant HCC surveillance and disease recurrence (Fig. 1). We propose that post-transplant surveillance strategies should be individualized based on prognostication scores and recurrence risk calculations. In those with high recurrence risk, screening should involve imaging and AFP levels every 3–6 months. If recurrence is diagnosed, the immunosuppression strategy should be re-evaluated, and the recurrence should be staged to distinguish oligo-recurrence from disseminated disease. For oligorecurrence, it is crucial to assess tumour location, disease burden, technical operative feasibility, residual organ function, and patient functional status to determine resectability. Where possible, surgical resection for intrahepatic and/or extrahepatic recurrence is recommended. Unresectable oligo-recurrence can be ablated or radiated. Multifocal intrahepatic disease can be treated with regional therapies, including TACE and Y90 for disease control. Disseminated disease is treated first-line with TKIs. There may also be a potential role for immunotherapy agents, including the PD-1 inhibitors, though the evidence is limited. With systemic therapy, care should be taken to monitor for side effects and graft dysfunction. Many clinical trials are currently recruiting towards developing and testing the efficacy and safety of new systemic therapies. Overall, there is a lack of high-evidence studies, and further research is required to develop better evidence-based treatment guidelines and newer drug treatment options for the management of post-transplant HCC recurrence.
Notes
Authors’ contributions
LR: Conception and design of project, literature review, write up of manuscript, critical revision, final approval of published version; TI: Conception and design of project, critical revision, final approval of published version; MPAWC: Conception and design of project, critical revision, final approval of published version; HM: Conception and design of project, critical revision, final approval of published version; GS: Conception and design of project, literature review, critical revision, final approval of published version
Conflicts of Interest
The authors have no conflicts to disclose.
Abbreviations
AASLD
American Association for the Study of Liver Diseases
AFP
alpha-fetoprotein
CNI
calcineurin inhibitors
CT
computed tomography
HCC
hepatocellular carcinoma
IQR
interquartile range
LT
liver transplantation
mTOR
mammalian target of rapamycin
mTORi
mammalian target of rapamycin inhibitors
OS
overall survival
PD-1
programmed cell death protein
RETREAT
Risk Estimation of Tumor Recurrence After Transplant
RFA
radiofrequency ablation
SBRT
stereotactic body radiation therapy
TACE
trans-arterial chemoembolization
TKI
tyrosine-kinase inhibitor
Y90
Yttrium-90 radioembolization