Tenofovir alafenamide treatment may not worsen the lipid profile of chronic hepatitis B patients: A propensity score-matched analysis
Article information
Abstract
Background/Aims
Tenofovir alafenamide (TAF) has shown less favorable effect on lipids compared to tenofovir disoproxil fumarate (TDF) in clinical trials. However, data regarding these outcomes in patients with chronic hepatitis B (CHB) are scarce. Therefore, this study aimed to evaluate the effect of TAF on the lipid in patients with CHB.
Methods
A total of 237 TAF-treated CHB patients compared with TDF, inactive CHB, and non-hepatitis B virus (HBV)-infected control groups using propensity score matching (PSM).
Results
Following PSM, each analysis was conducted on cohorts via the matching of 70:140 (TAF:TDF), 89:89 (TAF:inactive CHB), 140:560 (TAF:non-HBV infected control), and 368:1,472 (TDF:non-HBV-infected control). A significant decrease in the total cholesterol (TC) level was noted at 48 weeks in the TDF group compared to the TAF group (176.3±32.9 vs. 156.7±27.7, P<0.001) and the non-HBV-infected control group (175.0±29.5 vs. 156.2±28.3, P<0.001). However, no significant change in TC was observed in the TAF group and inactive CHB or non-HBV-infected control groups at 48 weeks. For the subgroup analyses of TAF vs. non-HBV-infected control subjects and inactive CHB patients whose detailed lipid profile information were available, no between-group differences in TC, low-density lipoprotein (LDL)-cholesterol, highdensity lipoprotein (HDL)-cholesterol, TC/HDL ratio, and LDL/HDL ratio were observed at 48 weeks.
Conclusions
TDF seems to have a lipid-lowering effect compared to the non-HBV-infected control and TAF-treated groups. However, in real practice, TAF might not worsen the lipid profiles of subjects compared to non-HBV-infected controls and patients with inactive CHB.
Graphical Abstract
INTRODUCTION
Hepatitis B virus (HBV) is the major cause of chronic hepatitis, cirrhosis, and hepatocellular carcinoma (HCC) [1]. Nucleos(t)ides (NAs) with potent viral suppression ability and high genetic barrier to resistance have been considered ideal drugs for achieving sustained viral suppression [2]. However, NA therapy does not usually result in the removal of the hepatitis B surface antigen, and virological relapse is common after cessation of treatment, leading to the need for indefinite therapy [3]. Long-term use of tenofovir disoproxil fumarate (TDF) has been associated with nephrotoxicity and bone mineral density reduction due to high circulating plasma levels of tenofovir (TFV) in some patients [4-6]. Tenofovir alafenamide (TAF) was designed to have greater plasma stability than TDF, allowing more efficient delivery of the active metabolite, TFV diphosphate, to hepatocytes compared to that of TDF [7,8]. Therefore, TAF has less impact on renal and bone safety [9]. Two large, multinational, phase III trials have demonstrated sustained antiviral efficacy of TAF that is non-inferior to TDF in patients with both hepatitis B e antigen (HBeAg)-positive and HBeAg-negative chronic HBV-infection [10,11]. The recently published 96-week data confirmed the efficacy outcomes observed at week 48 and at week 96 [12]. However, previous studies have indicated that TAF might be associated with a greater increase in total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), and triglyceride (TG) compared to TDF therapy [10-12]. This negative impact on the lipid profile of TAF may be associated with the risk of developing cardiovascular disease (CVD) [13]. Nevertheless, to date, no study has assessed the changes in lipid profile by TAF therapy in patients with chronic hepatitis B (CHB) in real-world practice. In order to clarify the effect of TAF on the lipid profile, it may be more suitable to compare TAF-treated patients to matched-healthy control subjects, rather than simply comparing them to matched-TDF group. The current study aimed to evaluate the effects of TAF on the lipid profile in a cohort of CHB patients, in comparison to TDF, non-HBV-infected control subjects, and patients with inactive CHB, using propensity score matching (PSM).
MATERIALS AND METHODS
This study was approved by the Institutional Review Board of Ulsan University Hospital (#IRB No. 06-2017-26), and the requirement for informed consent from patients was waived as patient records and information were anonymized prior to analysis.
Study population
We reviewed the electronic medical records of treatment-naïve CHB patients who were treated with TAF between March 2018 and February 2020 at Ulsan University Hospital, a tertiary referral center in South Korea. A total of 315 CHB patients treated with TAF were in the records. From January 2018 to January 2020, a total of 3,848 South Korean individuals were enrolled in this study as the control group. They had general health check-up at the Health Screening and Promotion Center in Ulsan University Hospital. A total of 900 patients treated with TDF in our CHB cohort [14] were also involved in this study as another control group. The inclusion criteria in this study for TAF group were as follows: 1) patients who were ≥18 years old with chronic HBV infection, 2) patients who had not received interferon therapy prior to TAF therapy, 3) patients with an estimated creatinine clearance ≥15 mL/min (by the Cockcroft-Gault method), and 4) patients who received treatment with TAF therapy for ≥48 weeks. We excluded patients with evidence of decompensation (i.e., clinical ascites, encephalopathy, or variceal hemorrhage), HCC, antibodies to hepatitis C virus, antibodies to human immunodeficiency virus (HIV), or autoimmune hepatitis. Additional criteria for exclusion were pregnancy, lactation, and habitual alcohol intake (>40 g/day ethanol). Moreover, patients who were treated with lipid-lowering agent within 48 weeks after the initiation of TAF therapy or those who started lipid-lowering agent intake during the observation period were also excluded. Seventy-eight patients who did not fulfill the inclusion criteria were excluded from this study, including 50 patients who received TAF treatment for less than 48 weeks. Sixteen patients were lost to follow-up. Lastly, 11 patients who initiated a lipid-lowering agent within 48 weeks of the TAF start date and one patient who initiated lipid-lowering agent during the observation period were excluded. Among patients who were treated with TDF, 14 who initiated a lipid-lowering agent within 48 weeks of the TAF start date and two patients who initiated lipid-lowering agent during the observation period, were excluded for the same reason regarding the lipid-lowering agent intake. The remaining 237 patients, who were treated with TAF and fulfilled all of the inclusion criteria, and 884 patients, who were treated with TDF, were included and matched with the control groups in this study. Information on the baseline patient characteristics and clinical outcomes were obtained from complete inpatient and outpatient medical records.
Follow-up evaluation
All patients underwent total blood counts, evaluation of biochemical and HBV-related virological markers, and HBV-DNA tests every 3–6 months during TAF and TDF therapy. The baseline lipid profile, mainly total TC, was evaluated for patients at 24 and 48 weeks after receiving TAF and TDF treatment. The healthy control group had a detailed lipid profile, including TC, LDL-C, high density lipoprotein-cholesterol (HDL-C), and TG, at baseline and 48 weeks. The authors of this study attempted to monitor all detailed lipid profiles for patients treated with TAF every 6 months in realclinical practice, since the clinical trials suggested that TAF may worsen lipid profiles.
Clinical outcomes and definitions
The primary endpoint was to investigate the changes of fasting TC during treatment in a cohort of CHB patients with TAF compared to TDF, inactive CHB, and non-HBV-infected control groups using PSM. The secondary endpoints included the following: 1) comparison of the LDL-C level, HDL-C level, TG level, TC/HDL-C ratio, and LDL-C/HDL-C ratio at baseline and 48 weeks in the non-HBV-infected healthy control groups or inactive CHB and TAF groups; and 2) comparison of the proportion of viral suppression and alanine transaminase (ALT) normalization in TAF and TDF groups. Liver cirrhosis was clinically defined when patients showed cirrhotic configuration of the liver following repeated imaging evaluations, such as ultrasonography or computerized tomography, thrombocytopenia (<150 K/µL), or splenomegaly and/or the presence of clinical signs of portal hypertension. Esophagogastroduodenoscopy was also performed annually to determine gastric or esophageal varices as evidence of portal hypertension. Diabetes mellitus was defined as the use of hypoglycemic agents including insulin, or a fasting glucose of ≥126 mg/dL. Hypertension was defined as blood pressure ≥140/90 mmHg or use of anti-hypertensive medications. Smoking status was classified as current smoker, ex-smoker, and non-smoker. Fatty liver disease was defined as diffuse increased hepatic parenchymal echogenicity, evaluated by abdomen ultrasonography. The virologic response at 12 months (VR 12) was defined as undetectable serum HBV DNA (<12 IU/mL) using the polymerase chain reaction assay for two consecutive measurements, 12 months after TAF or TDF therapy. Dyslipidemia was defined as TC ≥240 mg/dL, TG ≥150 mg/dL, or LDL-C ≥130 mg/dL in the fasting state [15] or use of lipid-lowering agent. Normal ALT was defined as ≤35 U/L for men and ≤25 U/L for women, which is the recommendation of the American Association for the Study of Liver Diseases [16].
Statistical analysis
Categorical variables are presented as frequencies and percentages. Continuous variables are presented as means with standard deviations (SDs) or medians with interquartile ranges. The Pearson chi-square test was employed for categorical variables, and a t-test (or Kruskal-Wallis test) was used to compare continuous numerical variables, as appropriate. To reduce the effect of test selection bias and potential confounding factors in this observational study, differences in baseline patient characteristics were adjusted using PSM. The propensity score-matched pairs were created by matching TAF, TDF, inactive CHB, and healthy control subjects on the logit of the propensity score, using calipers of width equal to 0.2 of the SD of the logit of the propensity score. We examined the similarity between TAF, TDF, and healthy control groups by calculating standardized differences for each of the baseline variables. All standardized differences for each baseline variable were <0.2 (20%). All reported P-values were two-sided. A P-value <0.05 was considered statistically significant. Data management, statistical analyses, and graph constructions were performed using Statistical Package for the Social Sciences (SPSS; version 24; SPSS Inc., Chicago, IL, USA), R-statistical software (version 3.3.1; R Foundation Inc.; http://cran.r-project.org/), and GraphPad-Prism (version 9-2; GraphPad Software, San Diego, CA, USA).
RESULTS
Baseline characteristics of the study population
We divided the control group (n=3,848) into inactive CHB patients (n=150) and non-HBV infected individuals (n=3,698), and then matched them to the TAF or TDF group. A total of 237 CHB patients treated with TAF and 884 CHB patients treated with TDF were analyzed. We compared the TAF group to the TDF and non-HBV-infected control groups, and also compared the TDF group to the non-HBV-infected control group. Patient characteristics were unbalanced between each group including the baseline HBV-DNA level. Therefore, we conducted 1:2 PSM for the TAF and TDF groups, 1:4 PSM for the TAF and non-HBV-infected control groups, 1:1 PSM for the TAF and inactive CHB groups, and 1:4 PSM for the TDF and non-HBV-infected control groups. Following PSM, each analysis was conducted on cohorts with the matching of 70:140 (TAF:TDF), 140:560 (TAF:non-HBV-infected control), 89:89 (TAF:inactive CHB), and 368:1,472 (TDF:non-HBV-infected control). The baseline characteristics of all patients treated with TAF, TDF, inactive CHB, and the non-HBV-infected control groups are summarized in Tables 1-3 and Supplementary Table 1. All parameters were balanced between the matched groups.

Baseline characteristics of the TAF and non-HBV-infected control groups and the propensity score-matched cohort
Comparison of TC changes in TAF and TDF groups
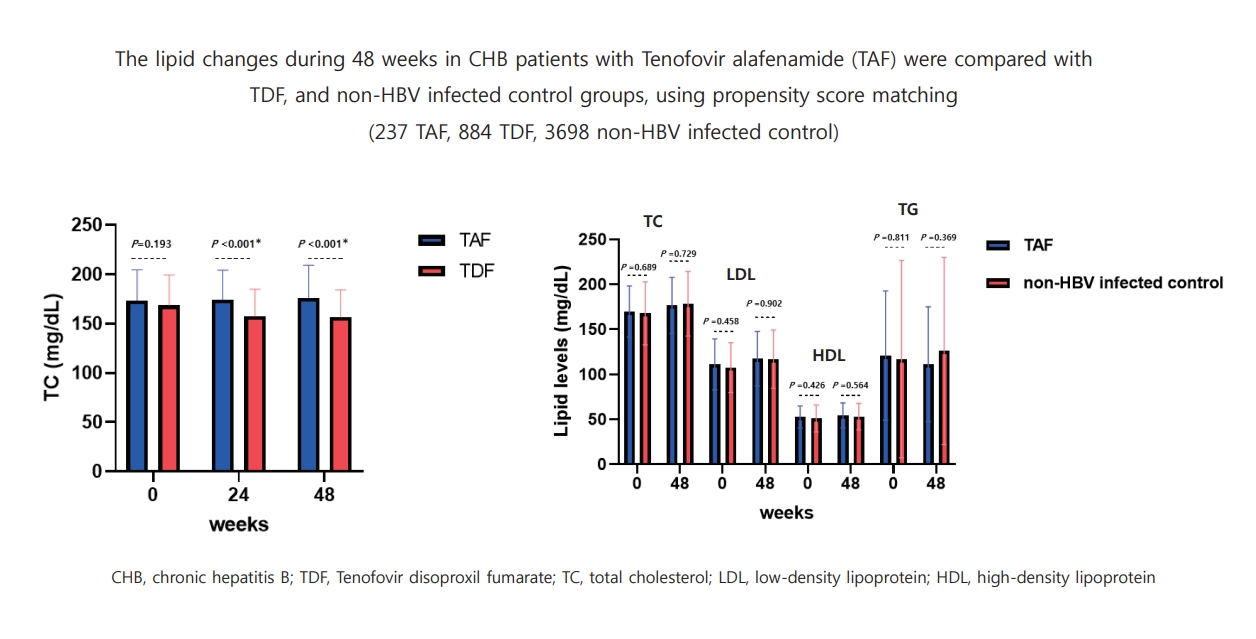
Following 1:2 PSM (Table 1), we compared TC at baseline and 48 weeks for the TAF and TDF groups. The mean±SD of the TC level at baseline in the TAF (n=70) and TDF groups (n=140) was not significantly different between groups at baseline (P=0.193); however, there was a significant difference at 48 weeks between groups (P<0.001) (Table 4; Supplementary Fig. 1C). Upon comparison of the changes in TC levels at 48 weeks, significant differences were observed between groups (-11.9±26.5, 2.9±25.2, P<0.001). At baseline and 48 weeks, the rate of patients with TC >240 mg/dL in the TAF and TDF groups was not significantly different (Table 4).
Clinical outcomes in patients treated with TDF and TAF in matched cohort
Comparison of baseline characteristics and clinical outcomes, including ALT normalization and VR 12, of the TDF and TAF groups are shown in Supplementary Table 2. The baseline characteristics were similar in both groups. The mean levels of systolic blood pressure, platelet count, serum albumin, serum bilirubin, prothrombin time, aspartate transaminase, ALT, and alpha-fetoprotein of the two groups were not significantly different. In addition, the proportion of patients with fatty liver disease was not different between groups (P=0.867). At 48 weeks, the mean±SD of serum creatinine was not different between groups (P=0.284). Notably, the TAF group had a higher proportion of patients with ALT normalization and VR12 compared to the TDF group at 48 weeks; however, there was no statistically significant difference between groups (76.7% vs. 85.9%, P=0.086; 81.5% vs. 89.5%, P=0.069, respectively) (Supplementary Table 2).
Comparison of TC changes in TAF and inactive CHB groups
Following 1:1 PSM (Table 2), we compared the TC levels at baseline and 48 weeks for the TAF and inactive CHB groups. The mean±SD of the TC level at baseline in the TAF (n=89) and inactive CHB groups (n=89) was not significantly different between groups at 48 weeks. Upon comparison of the changes in TC levels at 48 weeks, no significant differences were observed between groups. At baseline and 48 weeks, the rate of patients with TC >240 mg/dL in the TAF and inactive CHB groups were not significantly different (Table 4).
Comparison of detailed lipid profile changes in TAF and inactive CHB groups
Among the 1:1 PS-matched cohort, 56 patients each in the TAF and inactive CHB groups had detailed lipid profiles. At baseline, the mean±SD of the TC, LDL-C, HDL-C, TG, TC/HDL-C ratio, and LDL-C/HDL-C ratio in the TAF and inactive CHB groups had no significant differences (P=0.323, 0.683, 0.373, 0.085, 0.382, and 0.903, respectively). Upon comparing TC, LDL-C, HDL-C, TC/HDL-C ratio, and LDL-C/HDL-C ratio between the two groups at 48 weeks, no significant differences were observed between the TAF and inactive CHB groups (P=0.734, 0.997, 0.741, 0.172, 0.805, and 0.917, respectively) (Table 5; Fig. 1C, D).

Comparison of detailed lipid profile changes in the TAF group compared to the inactive CHB and non-HBV-infected control groups at baseline and 48 weeks
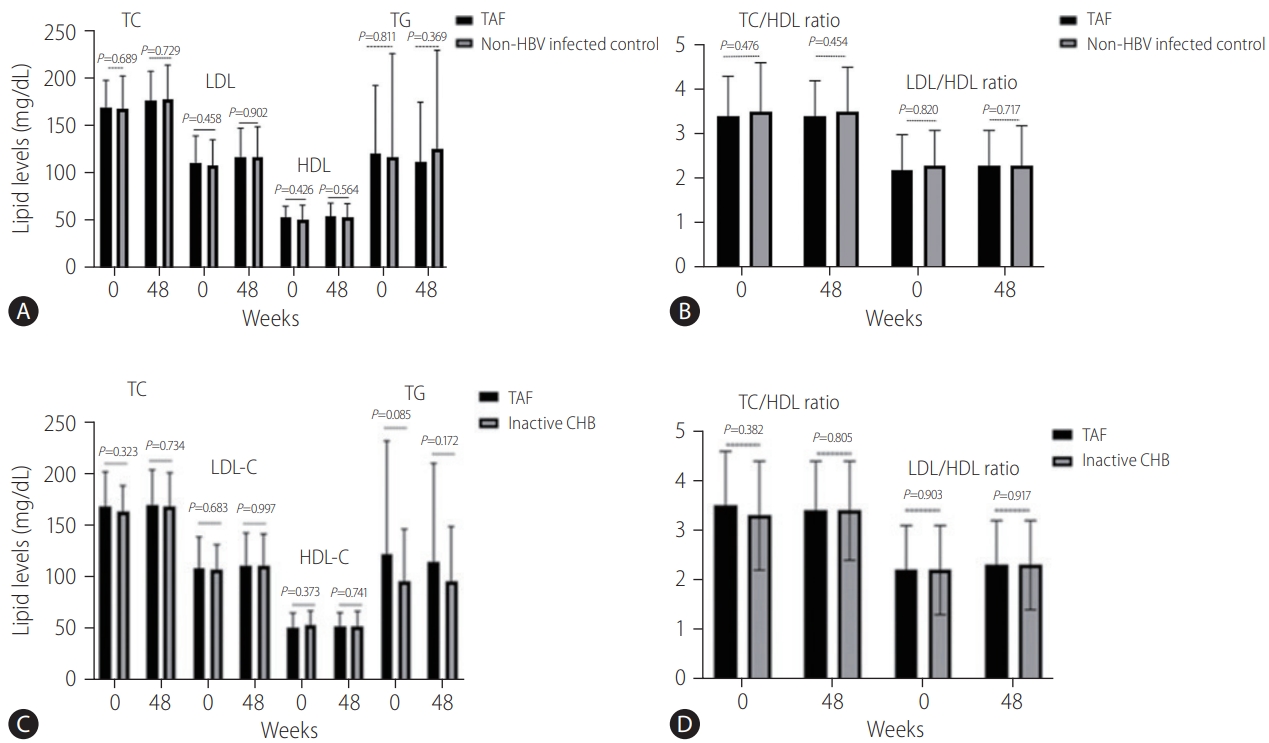
Comparison of all fasting lipid profiles, TC/HDL ratio, and LDL/HDL ratio in the TAF (n=45) and non-HBV-infected control groups (n=180). (A) All fasting lipid profile (TC, LDL, HDL, and TG) changes at baseline and 48 weeks in the TAF and non-HBV-infected control groups. (B) Comparison of the TC/HDL ratio and LDL/HDL ratio in the TAF and non-HBV-infected control groups. (C) All fasting lipid profile (TC, LDL, HDL, and TG) changes at baseline and 48 weeks in the TAF and inactive CHB groups. (D) Comparison of TC/HDL and LDL/HDL ratios in the TAF and inactive CHB groups. TC, total cholesterol; LDL, low-density lipoprotein; HDL, high density lipoprotein; TG, triglyceride; TAF, tenofovir alafenamide; CHB, chronic hepatitis B; HBV, hepatitis B virus.
Comparison of TC changes in TAF and non-HBV-infected control groups
Following 1:4 PSM, we compared the TC levels at baseline and 48 weeks of TAF and non-HBV-infected healthy control groups (Table 3). The mean±SD of the TC level at baseline in the TAF group (n=140) and the non-HBV infected healthy control group (n=560) was not significantly different at baseline and 48 weeks (Table 4; Supplementary Fig. 1A). Interestingly, an increase in TC level was observed in both groups at 48 weeks compared to the level at baseline (Supplementary Table 3). However, upon comparison of the changes in TC levels at 48 weeks, there was no significant difference between groups. At baseline and 48 weeks, the rate of patients with TC >240 mg/dL in the TAF and non-HBV-infected healthy control groups were not significantly different (Table 4).
Comparison of detailed lipid profile changes in TAF and non-HBV-infected control groups
In the PS-matched cohort, 45 TAF and 180 non-HBV-infected control subjects had detailed lipid profiles. During the follow-up period, TC and LDL-C increased in the TAF and non-HBV-infected control groups (Supplementary Table 3). However, the mean±SD of the TC, LDL-C, HDL-C, TG, TC/HDL-C ratio, and LDL-C/HDL-C ratio in the TAF and non-HBV-infected control groups had no significant differences at baseline. In addition, upon comparing TC, LDL-C, HDL-C, TG, TC/HDL-C ratio, and LDL-C/HDL-C ratio between two groups at 48 weeks, no significant differences were observed between the TAF and non-HBV-infected control groups (Table 5; Fig. 1A, B).
Comparison of TC changes in TDF and non-HBV-infected control groups
Following 1:4 PSM, we compared the TC level at baseline and 48 weeks of TDF and non-HBV-infected control groups (Supplementary Table 1). The mean±SD of the TC level at baseline in the non-HBV-infected control (n=1,472) and TDF groups (n=368) was 169.1±24.6 and 167.5±24.6, respectively (P=0.292); the mean±SD of the TC level at 48 weeks in the non-HBV-infected control group and TDF group was 175.0±29.5 and 156.2±28.3, respectively (P<0.001) (Supplementary Table 4, Supplementary Fig. 1B). Upon comparison of the changes in TC levels at 48 weeks, there was a significant difference between groups (5.8±23.4, -11.3±27.3, P<0.001). At baseline, the number of patients with TC >240 mg/dL in the control and TDF groups was nine (0.6%) and three (0.8%), respectively (P=1.000). However, at 48 weeks, the number of patients with TC >240 mg/dL in the control and TDF groups were 24 (1.6%) and one (0.3%), respectively (P=0.043) (Supplementary Table 4).
DISCUSSION
To the best of our knowledge, this is the first real-world study to evaluate the changes in lipid profile of CHB patients treated with TAF. Moreover, this is also the first study to compare the TC changes in CHB patients treated with TAF to those who underwent a general health examination, as well as inactive CHB patients and CHB patients treated with TDF. Our study showed that, compared to the non-HBV-infected controls and inactive CHB subjects who were not treated with anti-viral therapy, TAF might not worsen the lipid profiles of subjects in real clinical practice. Although no significant difference was observed for ALT normalization and VR 12, following the treatment of TAF and TDF groups, lipid-lowering effect of TDF was noted compared to healthy control subjects and TAF-treated patients.
Previous studies regarding HIV treatment have shown that the HIV treatment regimen, including TDF, has consistently been associated with lower lipid levels compared to the TAF-containing regimen in HIV treatment-naïve adults [17,18]. In HIV patients, CVD is an important cause of morbidity and mortality and is accompanied by lipid abnormalities [19]. Therefore, the occurrence or deterioration of dyslipidemia following HIV treatment has been a major concern [20,21]. However, there were conflicting results of TAF-associated lipid changes and CVD mortality estimation in real-life experiences of HIV-patients [11,22-27].
Regarding CHB treatment, randomized, double-blind, phase 3 clinical trial data indicated that patients who receive TAF therapy have a relatively higher fasting TC and LDL-C levels at 48 weeks and 96 weeks compared to the patients receiving TDF therapy [10-12]. However, there was no difference between groups in which patients initiated lipid-modifying agents at 48 weeks [11]. Moreover, the ratio of TC to HDL was not significantly different between the TAF and TDF groups at 96 weeks (P=0.14) [12], which was consistent with the results observed in HIV patients [13,25]. An integrated analysis of available data from clinical trials at week 48 showed that the proportion of patients with fasting TC and LDL levels above 300 mg/dL was 1% and 5%, respectively, in the TAF group and 0% and 1%, respectively, in the TDF group [10,11]. In the current study, the TC level of TDF group decreased significantly, which was apparently observed at 24 weeks after the TDF therapy. In addition, when comparing the TC levels at 48 weeks, the TDF group had a significantly lower TC level than compared to the TAF group. As previously published [12,25,26], we also observed a definite TC reduction in the TDF group compared to the non-HBV-infected healthy control group. Conversely, in the TAF and non-HBV-infected control groups, fasting TC level increased over the follow-up period in both groups; however, no significant changes in TC were observed at each time period (at baseline and 48 weeks) between the two groups. Notably, subgroup analysis of the TAF, inactive CHB, and non-HBV-infected control cohorts showed no significant change in all lipid parameters, including TC, LDL-C, and HDL-C. Moreover, neither the TC/HDL ratio nor the LDL/HDL ratio was different between groups. These results appear to be consistent with the explanation that TAF is lipid-neutral, as indicated in previous clinical trial [14]. It was speculated that the unique pharmacokinetics of TAF, which enables a high intracellular transfer compared with TDF and lowers plasma TFV concentration, may be a possible explanation. In addition to the results of the current study, a real-life cohort study in Canada demonstrated the TDF-related lipid-lowering effect compared to ETV treatment.28 Moreover, a propensity score-matched study recently revealed that TDF modulates lipid metabolism by upregulating hepatic CD36 via PPAR-α activation in vitro [29]. Therefore, the changes in fasting lipid levels may be more accurately reported, not as an adverse effect of TAF but as an effect of the absence of high plasma TFV concentrations in TAF-treated patients and TDF-related lipid modulation [18,22,28,29]. Although the clinical significance of these lipid changes is unclear, it is necessary to take it into consideration the aging of HBV-infected patients. As CHB patients grow older, they are more likely to have comorbidities and risk factors associated with CVD. Therefore, the results of the current study require robust exploration of appropriately designed studies and powerful cohorts with larger number of patients.
The strengths of this study included a large non-HBV-infected control group matched with TAF/TDF-treated patients, as well as a TAF group that was compared to the non-HBV-infected control, inactive CHB, and TDF groups using PSM analysis. This promoted increased statistical power and greater reliability of data. Nonetheless, this study had several limitations. First, we mainly compared the TC levels for the matched cohort, as fewer patients in the TDF cohort had detailed measurements of their lipid profile at both baseline and week 48. Therefore, we did not compare the LDL-C or TC/HDL ratio to the entire study population. However, as Korean population-based models with large prospective cohorts have suggested that an optimal TC level exists for the lowest CVD mortality [30-32], comparison of TC level is also considered clinically meaningful. Second, the observation period of this study may be relatively short for evaluating lipid changes with TAF. Third, the effect of TAF on patients with dyslipidemia is unknown, as patients who initiated lipid-lowering agents around the observation period were excluded from the current study. In addition, the study population did not include patients who were poorly controlled for dyslipidemia. Fourth, a liver biopsy was not performed on patients for the diagnosis of cirrhosis in the TAF and TDF groups; as a result, early detection of cirrhosis may have been missed. Fifth, we recognized the current medication and underlying disease of subjects in control groups based on the medical questionnaire of health screening center; however, due to the nature of the questionnaire, there might be some recall error among individuals. Lastly, this single-center study may be limited to the generalization of results.
In conclusion, this was the first real-world study to evaluate lipid changes in patients treated with TAF by comparing the TDF, inactive CHB, and non-HBV-infected control groups using PSM. The current study demonstrated that TAF might not worsen the lipid profile in patients with CHB. In addition, consistent with previous studies, this study showed a lipid-lowering effect of TDF compared to the TAF and non-HBV-infected control groups. In the future, studies with longer observation periods are required to confirm the lipid changes caused by TAF, and subsequent studies are also needed to assess the risk of CVD associated with TAF.
Notes
Authors’ contributions
Study coordination and design, data collection, data analysis, statistical analysis, writing and revision of the manuscript, and approval of the final version: J Jeong. Study coordination and design, data analysis, data supply, critical review of the manuscript, and approval of the final version: N.H.Park. Data analysis, statistical analysis and approval of the final version: E.J. Park. Data supply, critical review of the manuscript, and approval of the final version: J.W. Shin and S.W. Jung. All authors had full access to all data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Conflicts of Interest
The authors have no conflicts to disclose.
Acknowledgements
The authors would like to thank Big-data Center, Ulsan University Hospital.
SUPPLEMENTAL MATERIAL
Supplementary material is available at Clinical and Molecular Hepatology website (http://www.e-cmh.org).
Baseline characteristics of the TDF groups compared to the non-HBV infected control group and the propensity scorematched cohort
Clinical characteristics at baseline and 48-week clinical outcomes of TAF compared to TDF in the propensity scorematched cohort
Changes in all lipid profiles at baseline and 48 weeks in the TAF group compared to the non-HBV infected control subjects after propensity score matching
Comparison of TC changes in the TDF group compared to the non-HBV infected control group after propensity score matching
(A) Comparison of TC level at baseline to that at 48 weeks in the TAF (n=140) and non-HBV-infected control (n=560) groups. (B) Comparison of TC levels at baseline to that at 48 weeks in the TDF (n=368) and non-HBV-infected control groups (n=1,472). (C) Comparison of TC level at baseline to that measured at 24 and 48 weeks in the TAF (n=92) and TDF groups (n=276). TC, total cholesterol; TAF, tenofovir alafenamide; TDF, tenofovir disoproxil fumarate; HBV, hepatitis B virus.
Abbreviations
ALT
alanine transaminase
CHB
chronic hepatitis B
CVD
cardiovascular disease
HBeAg
hepatitis B e antigen
HBV
hepatitis B virus
HCC
hepatocellular carcinoma
HDL-C
high density lipoprotein-cholesterol
HIV
human immunodeficiency virus
LDL-C
low-density lipoprotein cholesterol
NAs
nucleos(t)ides
PSM
propensity score matching
SDs
standard deviations
TAF
tenofovir alafenamide
TC
total cholesterol
TDF
tenofovir disoproxil fumarate
TFV
tenofovir
TG
triglyceride
VR 12
virologic response at 12 months
References
Article information Continued
Notes
Study Highlights
• Relative deterioration of lipids may be observed in TAF-treated patients compared to TDF-treated patients.
• However, the details regarding lipid profiles, TC/HDL ratio, and LDL/HDL ratio were not significantly different between TAF-treated patients, inactive CHB patients, and non-HBV-infected control subjects.
• TAF treatment itself may not worsen the lipid profiles in real clinical practice.