Decreased vitamin D-binding protein level portends poor outcome in acute-on-chronic liver failure caused by hepatitis B virus
Article information
Abstract
Background/Aims
Acute-on-chronic liver failure (ACLF) is a catastrophic illness. Few studies investigated the prognostic value of vitamin D-binding protein (VDBP) for hepatitis B virus (HBV)-related ACLF (HBV-ACLF) resulted in conflicting results.
Methods
Two prospective HBV-ACLF cohorts (n=287 and n=119) were enrolled to assess and validate the prognostic performance of VDBP.
Results
VDBP levels in the non-survivors were significantly lower than in the survivors (P<0.001). Multivariate Cox regression demonstrated that VDBP was an independent prognostic factor for HBV-ACLF. The VDBP level at admission gradually decreased as the number of failed organs increased (P<0.001), and it was closely related to coagulation failure. The areas under the receiver operating characteristic curve (AUCs) of the Child-Pugh-VDBP and chronic liver failure-sequential organ failure assessment (CLIF–SOFA)-VDBP scores were significantly higher than those of Child-Pugh (P<0.001) and CLIF-SOFA (P=0.0013). The AUCs of model for end-stage liver disease (MELD)-VDBP were significantly higher than those of MELD (P= 0.0384) only in the case of cirrhotic HBV-ACLF patients. Similar results were validated using an external multicenter HBV-ACLF cohort. By longitudinal observation, the VDBP levels gradually increased in survivors (P=0.026) and gradually decreased in non-survivors (P<0.001). Additionally, the VDBP levels were found to be significantly decreased in the deterioration group (P=0.012) and tended to be decreased in the fluctuation group (P=0.055). In contrast, they showed a significant increase in the improvement group (P=0.036).
Conclusions
The VDBP was a promising prognostic biomarker for HBV-ACLF. Sequential measurement of circulating VDBP shows value for the monitoring of ACLF progression.
Graphical Abstract
INTRODUCTION
Acute-on-chronic liver failure (ACLF), characterized by acute decompensation of liver function and multi-organ failures on the basis of various chronic liver diseases, is a catastrophic end-stage syndrome with a high short-term mortality rate that can reach 30–50% at 28 days and 50–80% at 90 days [1-3]. Globally, 20–35% of patients with liver cirrhosis eventually progress to ACLF following various types of precipitation events [4]. In the Asian-Pacific region, chronic hepatitis B virus (HBV) infection is still one of main underlying etiologies for ACLF, while alarmingly, alcoholic steatohepatitis and metabolic associated fatty liver disease have also become important causes of ACLF in recent years [5].
Up to now, liver transplantation (LT) has been the only cure option for ACLF. However, LT resources are extremely scarce, and a considerable proportion of patients will not have the chance to receive LT due to the rapid progression to multi-organ failure or severe bacterial infection. In contrast, some patients may recover from the disorder and avoid LT, if timely and aggressive medical treatments are provided [6-8]. Therefore, precise stratification of patients and early prediction of ACLF outcomes are essential to save patients’ lives and avoid the wastage of precious LT resources. To address these challenges, there is an emerging interest in developing prognostic biomarkers for ACLF.
Liver-specific biomarkers have been advocated in recent years to measure the progression of liver failure and assist in its clinical management [9-12]. Vitamin D-binding protein (VDBP), synthesized and secreted by the liver, is a multifunctional protein involved in transporting vitamin D and its metabolites, scavenging actins released from necrotic cells, and performing nonspecific immune defense functions. It has been demonstrated that a low level of VDBP is associated with increased mortality in patients with liver failure [13-15]. However, the predictive performance of VDBP is distinct for different etiologies of liver failure [13,15,16]. Recently, liver-specific VDBP occurred in the HBV-related ACLF (HBV-ACLF) biomarker pool of our previous large-scale proteomics studies, suggesting that VDBP may be a biomarker to signal a poor prognosis in HBV-ACLF patients [9,10]. However, the limited number of studies that have aimed to assess the prognostic performance of VDBP on HBV-ACLF have demonstrated conflicting results [17,18]. It is therefore necessary to clarify the reliability of VDBP as a prognostic biomarker of HBV-ACLF. Additionally, it is hypothesized that VDBP levels are dynamic with evolving clinical features, so the sequential measurement of serial VDBP may assist clinicians in better managing ACLF.
We conducted a prospective study to assess the prognostic performance of VDBP alone and to test whether VDBP can be used as an adjunct indicator to improve current prognostic models in predicting the short-term outcomes of HBV-ACLF. The dynamic VDBP levels that are longitudinally correlated with the clinical course of HBV-ACLF were also investigated in the present study.
PATIENTS AND METHODS
Patients
A prospective, longitudinal HBV-ACLF cohort (n=287), enrolled from September 1, 2016 to September 1, 2018 at the First Affiliated Hospital of Zhejiang University, was used to assess the prognostic capability and to observe the dynamic alteration of VDBP in HBV-ACLF patients. New prognostic scoring models based on VDBP were also developed using this cohort. An external prospective multicenter HBV-ACLF cohort (validation cohort, n=119), enrolled at the First Affiliated Hospital of Nanchang University (n=23), the First Affiliated Hospital of Zhejiang University (n=37), the Xiangya Hospital of Central South University (n=33), and the Shulan Hospital of Hangzhou (n=26), was used to further validate the performance of VDBP-based scores (Supplementary Fig. 1). The study was conducted in compliance with the principles of the 1975 Declaration of Helsinki and was approved by the First Affiliated Hospital of Zhejiang University. Written informed consent was obtained from all patients or their legal representatives. Patients’ outcomes (survival or not) were followed up for 30 days after admission through the medical record system or by telephone.
Definition
HBV-ACLF was diagnosed at admission according to the criteria of the Asian Pacific Association for the Study of the Liver and was modified as previously described [9]. In brief, an acute hepatic insult manifesting as total bilirubin (TBil) ≥5 mg/dL and prothrombin time-international normalized ratio (PTINR) ≥1.5 within 4 weeks regardless of complicated by ascites or encephalopathy for patients with compensated cirrhotic or non-cirrhotic liver disease caused by HBV infection. In addition, patients with previously decompensated cirrhosis, who suffered an acute insult leading to sharply increased TBil (≥5 mg/dL) and PT-INR (≥1.5) within 4 weeks were also diagnosed as having ACLF. Exclusion criteria for HBV-ACLF patients are detailed in the Supplementary Materials.
The chronic liver failure-sequential organ failure assessment (CLIF-SOFA) score was used to diagnose the organ failure for HBV-ACLF patients [2]. Detailed criteria for failed organs can be seen in the Supplementary Data.
For longitudinal investigation, blood samples were collected at admission and on the 3rd, 7th, 10th, and 14th days during hospitalization whenever possible. Patients’ conditions were assessed by CLIF-SOFA score at admission and on the 14th day or at discharge [19]. Those with a ≥2-point decrease in CLIF-SOFA score were classified as the improvement group, patients with a ≥2-point increase in CLIF-SOFA score were classified as the deterioration group, and patients with an alteration of CLIF-SOFA between -1 and 1 during the observation were classified as the fluctuation group.
Data acquisition and laboratory detection
Data were obtained using a predesigned datasheet. The laboratory tests were performed with fasting blood samples at patients’ admission. Blood samples were collected at each time point and centrifuged at 3,500 rpm for 10 minutes, and were stored at -80°C. VDBP level was determined by commercial enzyme-linked immunosorbent assay (ELISA) kit (DVDBP0B; R&D, Minneapolis, MN, USA) in strict accordance with the manufacturer’s instructions.
Statistics
Statistical analyses were performed using Statistical Package for the Social Sciences (SPSS version 25.0; SPSS Inc., Chicago, IL, USA) and MedCalc (MedCalc Software, Ostend, Belgium). Continuous data are presented as the mean±standard deviation or the median with interquartile ranges. The student’s t-test, one-way analysis of variance, Mann-Whitney U test, and Kruskal-Wallis H test were used to compare the continuous data as appropriate. Categorical data were presented as numbers (percentages) and compared using chi-squared test or Fisher’s exact test. Univariate and multivariate Cox regression were used to identify independent prognostic factors for 30-day mortality of HBV-ACLF patients. Multivariate Cox regression was performed with the forward Wald method. The entry and removal probabilities were 0.05 and 0.10, respectively. The Wilcoxon test and the Greenhouse-Geisser method were used to compare the paired and repeated-measures data, respectively. The Spearman method was used to analyze the rank correlation, and Kaplan-Meier plots were used to compare the cumulative survival rates. Multivariate Cox regression analysis was performed to develop prognostic scores based on VDBP. The receiver operating characteristic (ROC) curves among scores were compared by Z-test using DeLong’s method. All tests were two-tailed, and P<0.05 was considered statistically significant.
RESULTS
Characteristics of HBV-ACLF patients at admission in the derivation cohort
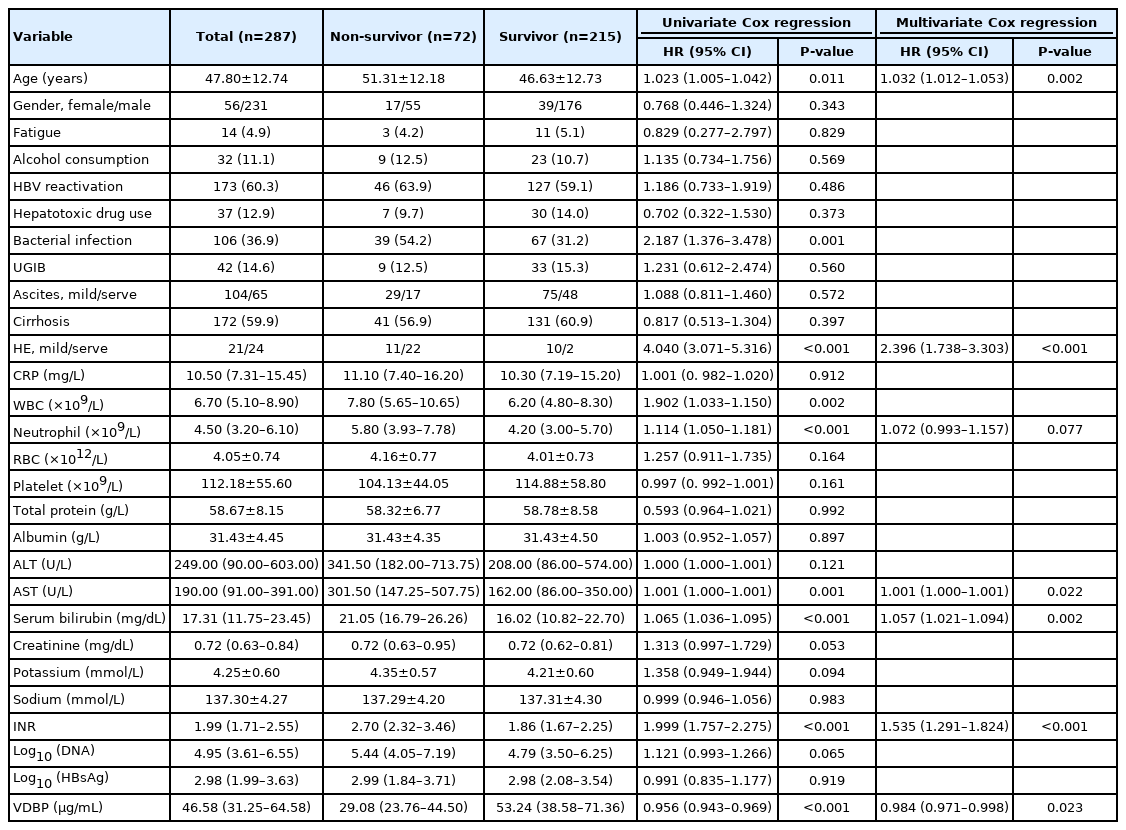
As shown in Table 1, of the 287 HBV-ACLF patients, 80.5% were men and 59.9% had underlying cirrhosis. The overall 30-day mortality rate of the HBV-ACLF patients was 25.1%. The most common precipitating events were HBV reactivation (60.3%), bacterial infection (36.9%), and upper gastrointestinal bleeding (14.6%). Comparing with ACLF patients precipitated by other events or unknown events, patients precipitated only by HBV reactivation are the youngest. In addition, patients precipitated only by HBV reactivation presented highest red blood cell counts, alanine aminotransferase, aspartate amino transferase (AST), PT-INR, serum HBV DNA levels, and HBsAg levels, but presented lowest TBil level (Supplementary Table 1). Univariate Cox regression indicated that the age of non-survivors was significantly older than that of survivors. The frequencies of infection and hepatic encephalopathy (HE) in non-survivors were significantly higher than in survivors. The white blood cell and neutrophil counts, and the levels of AST, TBil, and PT-INR were significantly higher in non-survivors than in survivors (Table 1).
Lower VDBP level at admission portended poor outcomes in HBV-ACLF patients
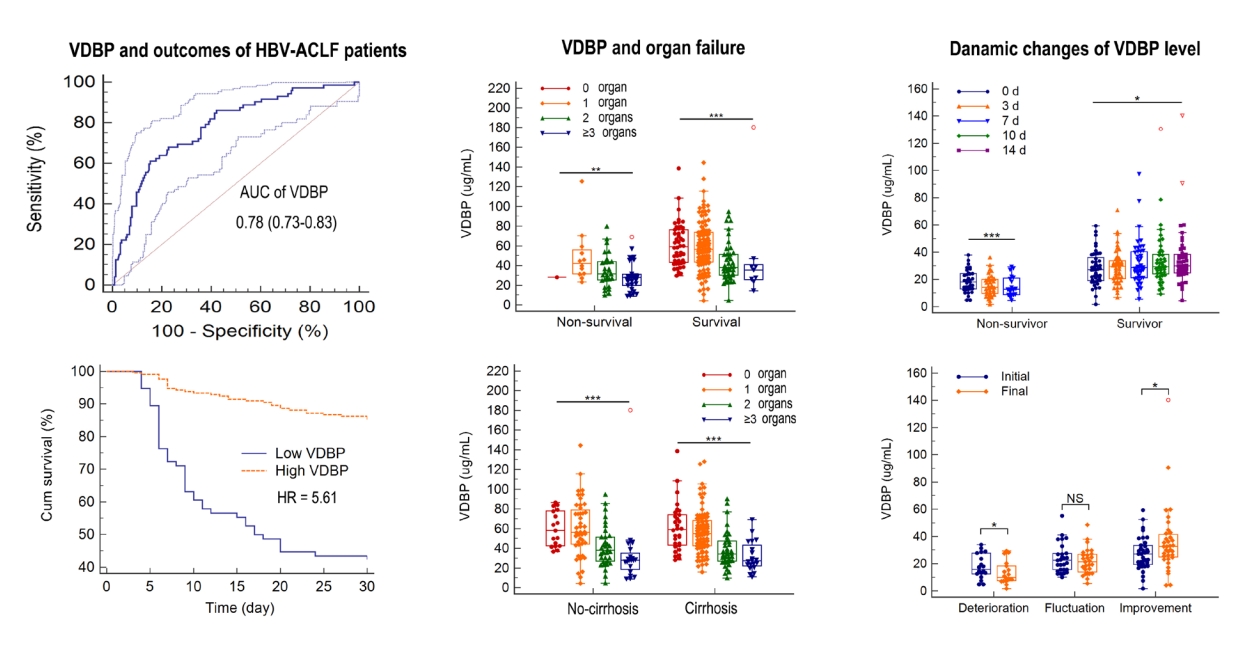
When compared at admission, VDBP levels in the non-survivors were significantly lower than in the survivors (29.08 [23.76–44.50] vs. 53.24 [38.58–71.36] mg/mL, P<0.001; Fig. 1A) during 30-day follow-up. When patients stratified ACLF by precipitating events, the results were similar for ACLF cases precipitated only by HBV reactivation or by other events (Supplementary Table 2). Multivariate Cox regression analysis demonstrated that VDBP, together with age, HE, neutrophil count, AST, TBil, and PT-INR were independent prognostic factors for HBV-ACLF (Table 1). When patients were stratified by the presence of cirrhosis, VDBP was still an independent prognostic factor in cirrhotic HBV-ACLF patients but not in non-cirrhotic HBV-ACLF patients (Supplementary Tables 3, 4).

Association of VDBP level at admission with HBV-ACLF prognosis. (A) VDBP distribution for patients with chronic HBV-ACLF in survivors and non-survivors. (B) The receiver operating characteristic curves of VDBP in prediction of 30-day mortality of HBV-ACLF patients. (C) Survival rates after 30 days of patients in the high- and low-VDBP groups. (D-F) The linear regression between VDBP and commonly used prognostic models. VDBP levels were compared using the Mann-Whitney U test, and Kaplan-Meier plots were compared by log-rank test. VDBP, vitamin D-binding protein; MELD, model for end-stage liver disease; CLIF-SOFA, chronic liver failure-sequential organ failure assessment; HBV-ACLF, hepatitis B virus-related acute-on-chronic liver failure. *P<0.001.
The area under the ROC curve (AUC) of VDBP levels for 30-day mortality was 0.780, with a sensitivity of 0.611 and a specificity of 0.847 at an optimal cut-off value of 31.68 mg/mL (Fig. 1B). When HBV-ACLF patients were divided into high-VDBP and low-VDBP sub-groups according to optimal cut-off value, we found that 43 patients (56.6%) with low-VDBP deceased, but 29 patients (13.7%) with high-VDBP deceased within 30 days. The cumulative survival rate of the low-VDBP group was significantly lower than that of the high-VDBP group (hazard ratio, 5.61; P<0.0001; Fig. 1C).
Furthermore, we assessed the association of VDBP level and disease severity at admission. VDBP levels were negatively correlated with Child-Pugh, model for end-stage liver disease (MELD), and CLIF-SOFA scores (all P<0.001, Fig. 1D-F).
Association of VDBP and organ failure.
As shown in Supplementary Table 5, in the first prospective longitudinal HBV-ACLF cohort (n=287), the most frequent organ failure was liver failure (73.9%), followed by coagulation (28.6%), respiratory (26.1%), and cerebral failure (7.7%). Circulation and kidney failure were uncommon in HBV-ACLF patients (2.1% and 1.4%, respectively). Most patients had single organ failure (44.9%), followed by two (24.7%), and more than two organ failures (13.9%). The VDBP level at admission gradually decreased with the increased number of failed organs (58.48 [42.47–76.54], 55.88 [42.79–71.67], 36.53 [26.88–50.99], and 27.51 [20.50–38.95] mg/mL, P<0.001; Fig. 2A). The VDBP levels also gradually decreased with the increased number of failed organs when patients were stratified by survival and non-survival outcomes (P<0.001 and P=0.005, respectively; Fig. 2B), or by status of underlying cirrhosis (both P<0.001, Fig. 2C). Additionally, as shown in Figure 3, the VDBP levels at admission were significantly lower in patients with liver failure than in those without liver failure (44.70 [28.93–61.87] vs. 54.57 [36.97–72.46], P=0.008). VDBP levels at admission were also lower in patients with (as opposed to without) coagulation failure (28.21 [21.76–36.98] vs. 54.42 [42.52–72.50], P<0.001), cerebral failure (27.70 [14.89–46.06] vs. 48.38 [32.70–66.74], P<0.001), respiratory failure (39.06 [27.77–57.28] vs. 49.60 [32.25–66.85], P=0.028), and circulation failure (25.70 [20.94–32.18] vs. 47.00 [31.55–65.45], P=0.006). When patients were stratified by clinical outcomes, VDBP levels were only significantly different in patients with and without coagulation failure, irrespective of them being survivors or non-survivors (Supplementary Fig. 2). When patients were stratified according to whether they had cirrhosis or not, the VDBP level in HBV-ACLF patients with coagulation failure was significantly lower than in those without, irrespective of whether they were cirrhotic or not. The results were similar for patients with and without cerebral failure. Significantly lower VDBP levels were observed in patients with liver failure than in those without for cirrhotic HBV-ACLF but not for non-cirrhotic HBV-ACLF. No significant VDBP difference was observed between patients with respiratory failure and those without in both cirrhotic and non-cirrhotic HBV-ACLF patients (Supplementary Fig. 3).
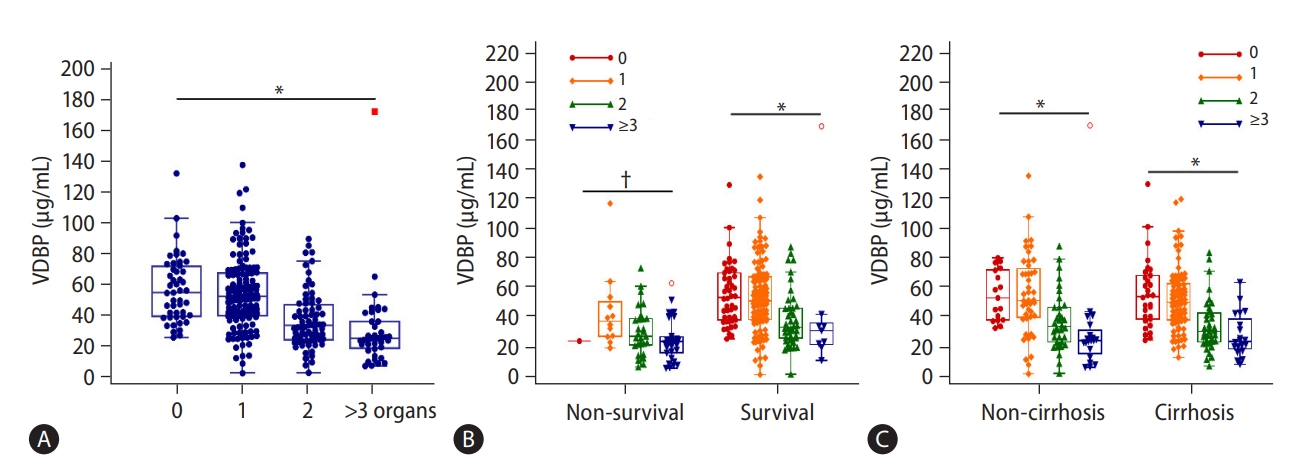
Association of VDBP and the number of organ failures. VDBP distribution with increased numbers of organ failure (A). VDBP distribution with increased numbers of organ failure in ACLF survivors and non-survivors (B). VDBP distribution with increased numbers of organ failure in ACLF patients with and without cirrhosis (C). Rank correlation was analyzed by the Spearman method. VDBP, vitamin D-binding protein; ACLF, acute-on-chronic liver failure. *P<0.001. †P<0.01.
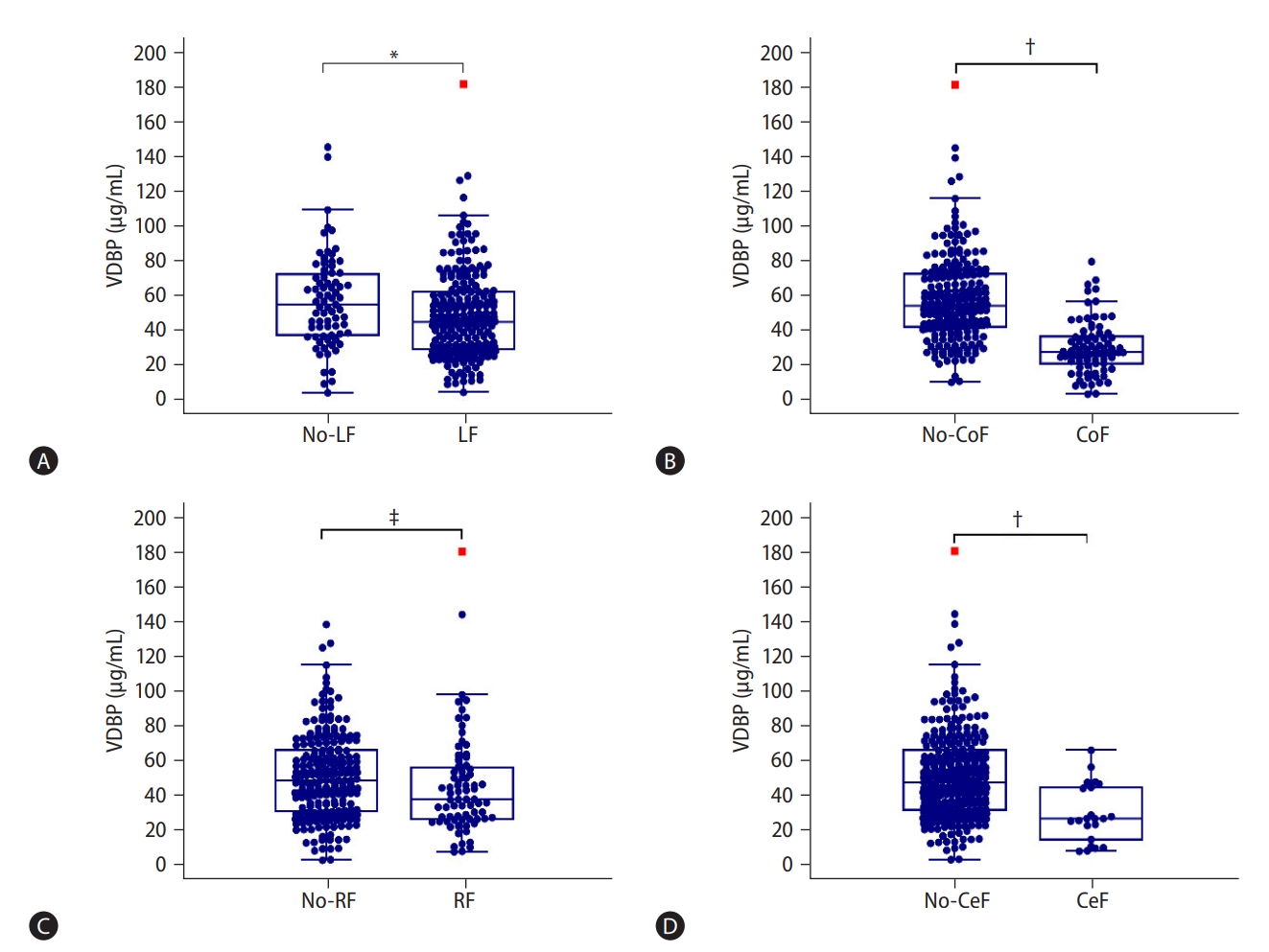
Alterations of VDBP levels according to organ failure. VDBP distribution of HBV-ACLF patients with and without (A) liver failure, (B) coagulation failure, (C) respiratory failure, and (D) cerebral failure. The associations of VDBP with circulation and kidney failures were not shown due to their low frequencies. VDBP levels were compared using the Mann-Whitney U test. VDBP, vitamin D-binding protein; LF, liver failure; CoF, coagulation failure; RF, respiratory failure; CeF, cerebral failure; HBV-ACLF, hepatitis B virus-related acute-on-chronic liver failure. *P<0.01. †P<0.001. ‡P<0.05.
Then we observed the AUC of VDBP for predicting organ failures. As shown in Supplementary Figure 4, excellent predictive capacity was observed in predicting coagulation failure, with an AUC of 0.851 (0.804–0.890). A moderate AUC of 0.767 (0.713–0.814) was observed for predicting cerebral failure. Unsatisfactory predictive capacities were observed in predicting both liver failure and respiratory failure (AUC, 0.603 [0.543–0.660] and 0.586 [0.526–0.643], respectively).
VDBP enhanced the predictive capacities of current models for ACLF
We further investigated the prognostic performance of VDBP in conjunction with commonly used clinical models in predicting 30-day mortality of HBV-ACLF. Three novel prognostic models were developed by the multivariate Cox regression analysis: Child-Pugh-VDBP = 4.7 × Ln(Child-Pugh) – 1.0 × Ln(VDBP); MELD-VDBP = 0.2 × MELD–0.9 × Ln(VDBP); CLIF-SOFA-VDBP = 3.7 × Ln (CLIF-SOFA) – 0.7 × Ln(VDBP).
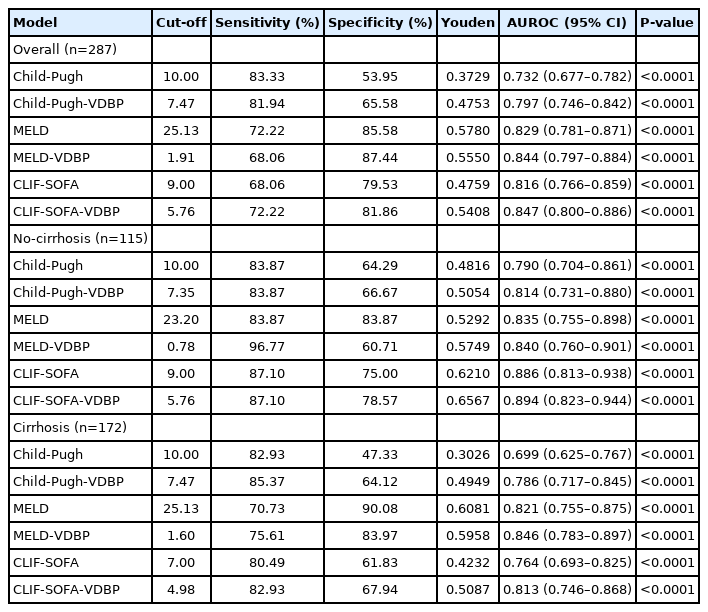
As shown in Figure 4A-C, for patients overall, the AUC of Child-Pugh-VDBP was significantly higher than that of Child-Pugh (0.797 [95% CI, 0.746–0.842] vs. 0.732 [95% CI, 0.677–0.782], P<0.001). The AUC of CLIF-SOFA-VDBP was also significantly higher than that of CLIF-SOFA (0.847 [95% CI, 0.800–0.886] vs. 0.816 [95% CI, 0.766–0.859], P=0.0013). The AUC of MELD-VDBP was slightly higher than that of MELD score, but the difference was not significant (0.846 [95% CI, 0.799–0.886] vs. 0.829 [95% CI, 0.781–0.871], P=0.199). For HBV-ACLF patients with cirrhosis, the AUC of Child-Pugh-VDBP was significantly higher than that of Child-Pugh score (0.786 [95% CI, 0.717–0.845] vs. 0.699 [95% CI, 0.625–0.767], P=0.001; Fig. 4D). The AUCs of MELD-VDBP and CLIF-SOFA-VDBP were also significantly higher than those of MELD score (0.846 [95% CI, 0.783–0.897] vs. 0.821 [95% CI, 0.755–0.875], P=0.038) and CLIF-SOFA (0.813 [95% CI, 0.746–0.868] vs. 0.764 [95% CI, 0.693–0.825], P<0.001; Fig. 4E, F) respectively. For HBV-ACLF patients without cirrhosis, as shown in Supplementary Figure 5, no significant AUC difference was observed between Child-Pugh-VDBP and Child-Pugh score (P=0.434), between Child-Pugh-MELD and MELD score (P=0.820), or between CLIF-SOFA-VDBP and CLIF-SOFA (P=0.576). The detailed parameters for the ROC curves of the models are shown in Table 2.
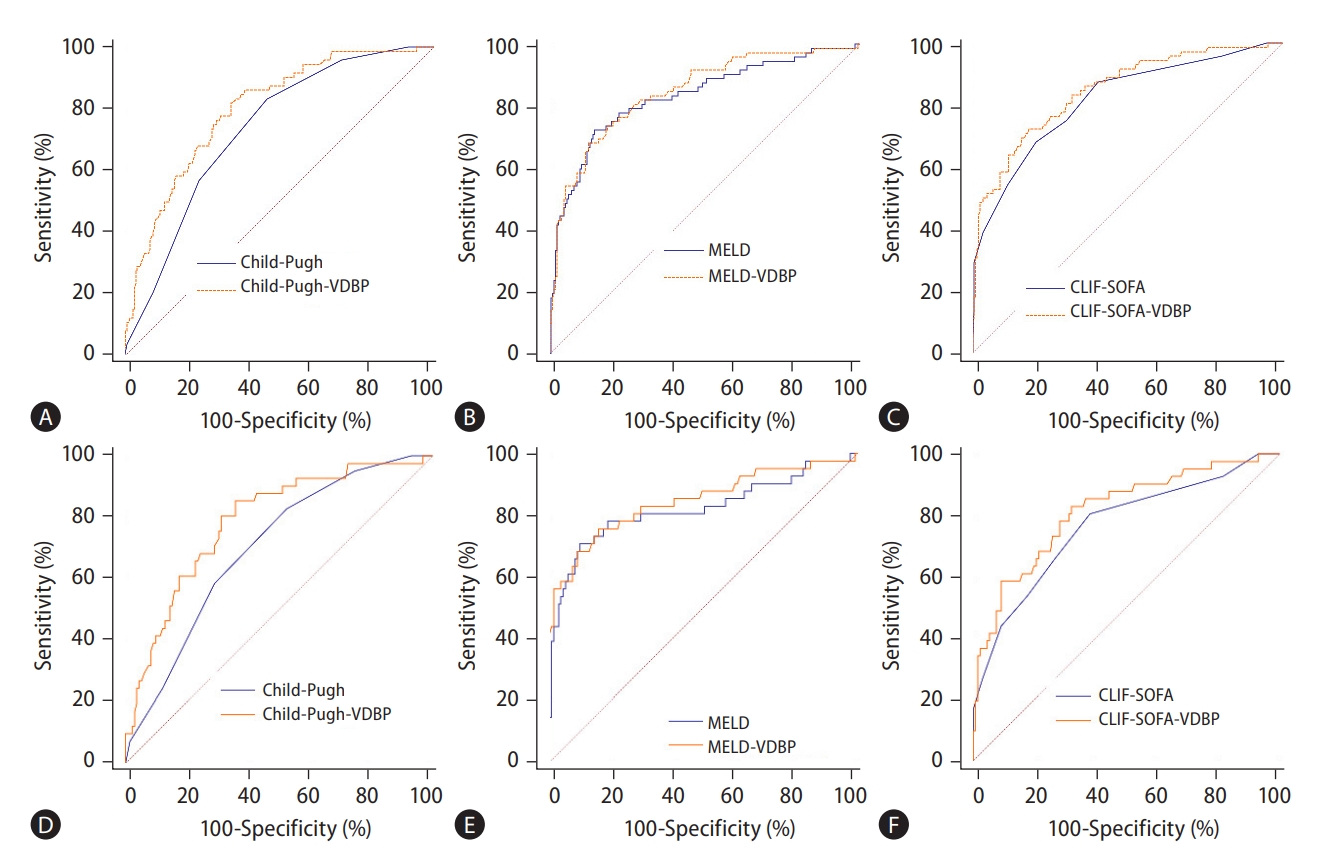
The prognostic performance of VDBP-based models in predicting 30-day mortality of HBV-ACLF patients in the derivation cohort. The prognostic performance of VDBP-based models for overall (A-C) and cirrhotic (D-F) HBV-ACLF patients, respectively. VDBP, vitamin D-binding protein; MELD, model for end-stage liver disease; CLIF-SOFA, chronic liver failure-sequential organ failure assessment; HBV-ACLF, hepatitis B virus-related acute-on-chronic liver failure.
Validation of the prognostic performance of VDBP-based models
The present study further enrolled a multicenter cohort to validate the prognostic performance of VDBP-based models. The characteristics at admission of the derivation and validation cohorts are shown in Supplementary Table 6. To predict poor outcome in all patients, the AUC of Child-Pugh-VDBP was significantly higher than that of Child-Pugh (P<0.001, Fig. 5A), and the AUCs of MELD-VDBP and CLIF-SOFA-VDBP tended to be higher than those of MELD and CLIF-SOFA scores, respectively (P=0.051 and P=0.061, Fig. 5B, C). For HBV-ACLF patients with cirrhosis, the AUCs of Child-Pugh-VDBP and MELD-VDBP were significantly higher than those of Child-Pugh and MELD scores, respectively (P<0.001 and P=0.025, Fig. 5D, E). The AUC of CLIF-SOFA-VDBP tended to be higher than that of CLIF-SOFA score (P=0.052, Fig. 5F). The detailed parameters for the ROC curves of the models are shown in Supplementary Table 7.

The prognostic performance of VDBP-based models in predicting 30-day mortality of HBV-ACLF patients in the external validation cohort. The prognostic performance of VDBP-based models for overall (A-C) and HBV-ACLF patients with cirrhosis (D-F), respectively. The prognostic performance of VDBP-based models for HBV-ACLF patients without cirrhosis was not analyzed because the sample size was too small to draw reliable conclusions. VDBP, vitamin D-binding protein; MELD, model for end-stage liver disease; CLIF-SOFA, chronic liver failure-sequential organ failure assessment; HBV-ACLF, hepatitis B virus-related acute-on-chronic liver failure.
Dynamic VDBP levels with clinical course
Among the HBV-ACLF patients, 36 non-survivors with available 7-day follow-up serum samples and 48 survivors with available 14-day follow-up serum samples were enrolled to observe the dynamic changes of VDBP during the patients’ hospitalization. As shown in Figure 6A, VDBP levels gradually increased in survivors (P=0.026) and gradually decreased in non-survivors (P<0.001). Among the 84 patients, 18 suffered a deterioration in condition (CLIF-SOFA score increased ≥2), 36 experienced improvement (CLIF-SOFA score decreased ≥2), and 30 experienced fluctuations (CLIF-SOFA score changed <2) during their hospitalization. As shown in Figure 6B, compared with those at admission, the VDBP levels at the final assessment (14 days after admission or discharge) were found to have significantly decreased in the deterioration group (32.00 [23.99–56.03] vs. 19.87 [15.10–37.57] mg/mL, P=0.012) but significantly increased in the improvement group (54.42 [interquartile range, 38.65–67.06] vs. 65.35 [interquartile range, 49.54–84.46] mg/mL, P=0.036). The VDBP levels at the final assessment decreased slightly, but not significantly, in the fluctuation group (P=0.055).
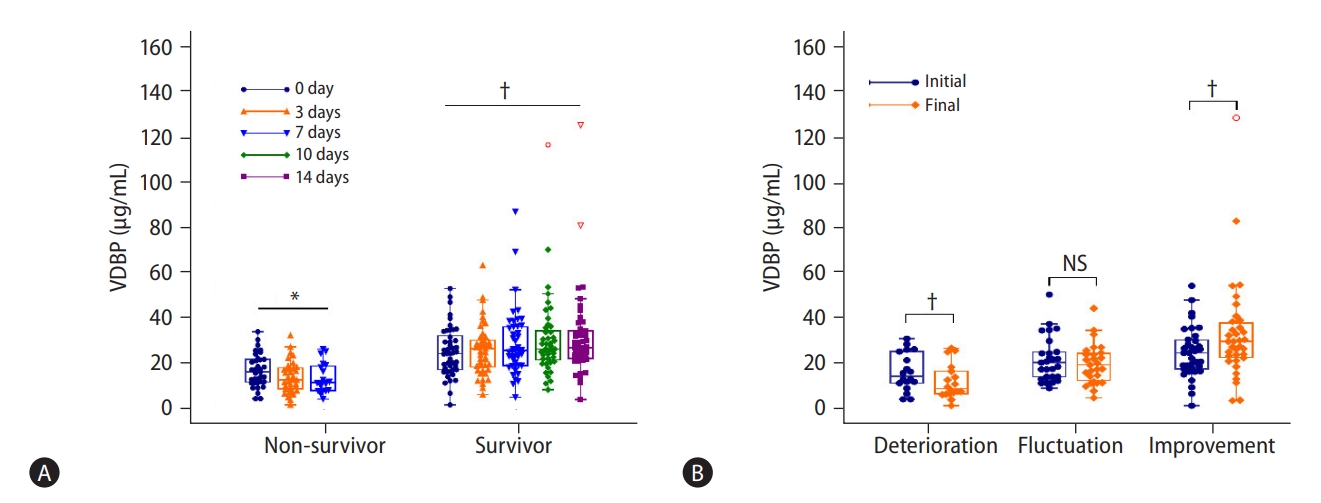
Longitudinal changes of VDBP levels according to clinical course. (A) Dynamic changes of VDBP levels in HBV-ACLF survivors and non-survivors by longitudinal observation during hospitalization. (B) Changes in VDBP levels between the initial (admission) and final (14 days after admission or discharge) assessments. Repeated-measures data were analyzed with the Greenhouse-Geisser method, and paired data were analyzed with Wilcoxon tests. VDBP, vitamin D-binding protein; NS, not significant; HBV-ACLF, hepatitis B virus-related acute-on-chronic liver failure. *P<0.001. †P<0.05.
DISCUSSION
Actin, the predominant intracellular protein, is released in large amounts from the necrotic liver cells of patients with liver failure. Free actin may cause thrombosis in vascular system and lead to local tissue ischemia and subsequent multiple organ dysfunction [20,21]. VDBP plays a crucial role in scavenging circulating actin and thus preventing organ injury. We speculate that a lower level of circulating VDBP in patients with liver failure is due primarily to the excessive consumption of VDBP in order to quench free actin derived from necrotic hepatocytes. Hence, dynamic monitoring of the circulating VDBP level facilitates the assessment of liver failure severity. The studies that have investigated the prognostic value of VDBP in terms of HBV-ACLF are quite limited, and their results are not conclusive. Yin and Chen [17] reported that lower VDBP is an effective indicator for predicting outcome and can be used for risk stratification. However, Liu et al. [18] deemed that VDBP is not a suitable indicator for HBV-ACLF prognosis. This discrepancy may be attributable to the small sample sizes of the two studies. The existence of multiple forms of VDBP may also explain the contradictory results. Moreover, because VDBP contains three albumin-like domains, the highly abundant albumin in serum samples could interfere with VDBP immunoassays, leading to unreliable measurements, particularly for ACLF patients with low VDBP levels. Thus, the present study represented the largest prospective cohort reported so far and aimed to assess the prognostic performance of total VDBP in predicting the outcome of HBV-ACLF using ELISA assay free of albumin/actin interference. The study found that VDBP is an independent factor for HBV-ACLF prognosis, and lower VDBP levels portended a poor outcome for patients. The optimal cutoff value of 31.68 μg/mL for AUC of VDBP carried the best prognostic information in predicting HBV-ACLF outcome, which was almost identical to previous studies based on acute liver failure cohorts [22,23]. This study also investigated the dynamic alteration of VDBP levels by means of 14-day longitudinal observation, and found that VDBP levels gradually increased in survivors but gradually decreased in non-survivors after a 14-day longitudinal observation during hospitalization. Moreover, the VDBP level significantly increased in patients whose condition improved but significantly decreased in subjects whose condition deteriorated. These results robustly demonstrated that VDBP was a promising prognostic biomarker for HBV-ACLF patients. The VDBP level in circulation is determined by its synthesis and consumption. It is reported that the hepatic synthesis of VDBP is increased in liver failure for its high priority [24]; however, obviously decreased VDBP levels were observed in HBV-ACLF non-survivors, indicating that more profound VDBP consumption occurred in those patients, either because of actin complex formation and then removal from circulation or because of consumption as part of immune responses.
Organ failure, especially subsequently developed extra-hepatic multisystem organ failure, is a critical development in ACLF and leads to high mortality of ACLF patients [25]. PT-INR, an indicator of failed liver function, is also a marker for subsequent coagulation failure. Lu et al. [26] recently demonstrated that a gradually increasing PT-INR index was associated with poor survival and indicated the need for fast-track liver transplant. Consistent with that result, the present study found that coagulation failure was also closely correlated with VDBP level when patients were stratified by clinical outcome. The underlying mechanism by which decreased VDBP levels are connected to failed organs may be associated with clearance of microthrombosis in microvascular system caused by excessive actin, which is an important initiating and exacerbating event in multiple organ failure [27].
Different predictive models have been established to determine the outcomes of patients with ACLF and to facilitate organ allocation for LT. Child-Pugh and MELD scores are the most used scores for evaluating the severity of end-stage liver disease. CLIF-SOFA, a score used to evaluate organ failures, is also widely used to predict the outcomes of ACLF patients. On top of these existing scores, we developed Child-Pugh-VDBP and CLIF-SOFA-VDBP, the prognostic performances of which were significantly higher than those of Child-Pugh and CLIF-SOFA scores, respectively. For HBV-ACLF patients with cirrhosis, VDBP also significantly enhanced the prognostic performance of MELD score. Thus, these VDBP-augmented scores showed great potential in facilitating the clinical management of HBV-ACLF.
There were some limitations in our study. First, the impact of VDBP combined with actin (VDBP-actin complexes) on HBV-ACLF prognosis was not investigated in this study, as accurate and reliable measurement of VDBP-actin complexes was not yet available. However, the VDBP-actin complexes are rapidly cleared (usually within 30 minutes) by the liver and other organs once formed [28], and a previous study demonstrated that the VDBP-actin complexes were not useful in predicting the outcome of liver failure [24]. Second, the usefulness of VDBP as a prognostic biomarker for HBV-ACLF patients was confirmed by this study. Further studies are required to confirm whether the results can be extended to ACLF caused by other etiologies.
In conclusion, the present study enrolled the largest prospective HBV-ACLF cohort reported to confirmed the prognostic value of VDBP in predicting the outcome of HBV-ACLF from four aspects: (1) this study revealed a profoundly decreased VDBP level in non-survivors and demonstrated VDBP as an independent prognostic factor for HBV-ACLF patients; (2) this study investigated the negative correlations between VDBP and ACLF-associated failed organs; (3) this study observed the enhancement effect of VDBP on current ACLF prognostic models; and (4) more importantly, this study revealed the dynamic alteration of VDBP levels with the clinical course of HBV-ACLF. We believe that VDBP shows great value in facilitating clinical decision-making for HBV-ACLF management.
Notes
Authors’ contributions
Zeyu Sun was the guarantor of the submission. Daxian Wu, Zeyu Sun, and Xiaoyu Cheng designed the study. Daxian Wu, Qunfang Rao, Zhongyang Xie, Xiaoqing Zhu, Jian Wu, Hainv Gao, Jingyu Zhang, and Zhouhua Hou contributed the data acquisition. Daxian Wu, Qunfang Rao, and Jingyu Zhang performed the serological detection. Daxian Wu, Yuanmei Che, and Zeyu Sun analyzed and interpreted the data. Daxian Wu drafted the manuscript, and Zeyu Sun provided critical revision of the manuscript. All authors approved the final version of the manuscript.
Conflicts of Interest
The authors have no conflicts to disclose.
Acknowledgements
This work was supported by the National Natural Science Foundation of China [82000601], the National Key Research and Development Program (2017YFC1200204), and the Research Project of Jinan Microecological Biomedicine Shandong Laboratory (JNL-2022030C). The funders had no role in the collection, analysis or interpretation of the data; in the writing of the report; or in the decision to submit the article for publication.
SUPPLEMENTAL MATERIAL
Supplementary material is available at Clinical and Molecular Hepatology website (http://www.e-cmh.org).
Patients’ characteristics of all subjects
Characteristics on admission among different precipitating events in HBV-ACLF patients of the derivation cohort
Comparison of characteristics on admission between non-survivors and survivors in HBV-ACLF patients of the derivation cohort stratified by precipitating events
Comparison of characteristics on admission between non-survivors and survivors in HBV-ACLF patients without cirrhosis of the derivation cohort
Comparison of characteristics on admission between non-survivors and survivors in HBV-ACLF patients with cirrhosis of the derivation cohort
Organ failure on admission according to survival in the derivation cohort
Characteristics at admission of the derivation and validation cohorts
Performance of the various scores for predicting the prognosis of patients with HBV-ACLF in the validation cohort
Flow chart of patient enrollment. HBV-ACLF, hepatitis B virus-related acute-on-chronic liver failure; HIV, human immunodeficiency virus.
Alterations of VDBP levels according to organ failure stratified by clinical outcomes. VDBP distribution of HBV-ACLF patients with and without (A) liver failure, (B) coagulation failure, (C) respiratory failure, and (D) cerebral failure. The association of VDBP and circulation and kidney failures were not shown for their low frequencies. VDBP levels were compared using the Mann-Whitney U test. VDBP, vitamin D-binding protein; LF, liver failure; NS, not significant; CoF, coagulation failure; RF, respiratory failure; CeF, cerebral failure; HBV-ACLF, hepatitis B virus-related acute-on-chronic liver failure. *P<0.05. †P<0.001.
Alterations of VDBP levels according to organ failure stratified by underlying cirrhosis. VDBP distribution of HBV-ACLF patients with and without (A) liver failure, (B) coagulation failure, (C) respiratory failure, and (D) cerebral failure. The association of VDBP and circulation and kidney failures were not shown for their low frequencies. VDBP levels were compared using the Mann-Whitney U test. VDBP, vitamin D-binding protein; NS, not significant; LF, liver failure; CoF, coagulation failure; RF, respiratory failure; CeF, cerebral failure; HBV-ACLF, hepatitis B virus-related acute-on-chronic liver failure. *P<0.05. †P<0.001. ‡P<0.01.
The prognostic performances of VDBP in predicting organ failure in the derivation cohort. The prognostic performances of VDBP in predicting liver failure (A), coagulation failure (B), respiratory failure (C), and cerebral failure (D) of ACLF patients. VDBP, vitamin D-binding protein; ACLF, acute-on-chronic liver failure.
(A-C) The prognostic performance of VDBP based models in predicting 30-day mortality of HBV-ACLF patients without cirrhosis in derivation cohort. VDBP, vitamin D-binding protein; MELD, model for end-stage liver disease; CLIF-SOFA, chronic liver failure–sequential organ failure assessment; HBV-ACLF, hepatitis B virus-related acute-on-chronic liver failure.
Abbreviations
ACLF
acute-on-chronic liver failure
AST
aspartate aminotransferase
AUC
area under the ROC curve
CLIF-SOFA
chronic liver failure–sequential organ failure assessment
ELISA
enzyme-linked immunosorbent assay
HBV-ACLF
HBV-related ACLF
HBV
hepatitis B virus
HE
hepatic encephalopathy
PT-INR
prothrombin time-international normalized ratio
LT
liver transplantation
MELD
model for end-stage liver disease
RBC
red blood cell
TBil
total bilirubin
VDBP
vitamin D-binding protein
References
Article information Continued
Notes
Study Highlights
• This work aimed to clarify the prognostic value of VDBP for HBV-ACLF by two prospective HBV-ACLF cohorts with large sample size.
• Lower VDBP level at admission portended poor outcome of HBV-ACLF patients and VDBP improved the predictive capacities of current models for HBV-ACLF prognosis.
• The VDBP level was gradually decreased with the increased number of failed organs and changed dynamically with the clinical course.