| Clin Mol Hepatol > Volume 23(1); 2017 > Article |
|
ABSTRACT
Hepatogastric fistula following a pyogenic liver abscess is extremely rare, and only a handful of cases have been reported. An 88-year-old female presented with generalized weakness, fever and chills. An abdominal computed tomography scan revealed a 5cm-sized hypodense lesion with internal septa in the left lateral section of the liver. Due to initial suspicion of early liver abscess, she was treated with empirical intravenous antibiotics. Initially, aspiration or drainage of the liver abscess was not performed due to immature lesion characteristics. An ultrasonography-guided percutaneous drainage of the liver abscess was performed 17 days after hospitalization due to a more mature lesion appearance on follow-up imaging. On tubography, contrast media leakage through the fistulous tract was visualized. Surgical management was performed, and she was discharged 2 weeks after surgery.
Although several complications of liver abscess have been reported, cases of hepatogastric fistula secondary to liver abscess are rare [1]. There are no established guidelines for the diagnosis and management of this complication. Although surgical management is a definitive treatment, conservative management has also shown clinical improvement in several reports [2-4]. We present a case of liver abscess complicated by hepatogastric fistula that was successfully managed with surgery.
An 88-year-old woman without relevant medical history visited the emergency room because of generalized weakness associated with fever and chills over the prior 3 days. She reported no history of cigarette smoking or alcohol consumption. Her body temperature was 38.7┬░C. On examination there was tenderness in the right upper abdomen. Laboratory tests revealed a white blood cell (WBC) count of 11,380/╬╝L, with 87% neutrophils, and a platelet level of 25,000/╬╝L. The patientŌĆÖs blood chemistry profile was as follows: fasting glucose, 103 mg/dL; total protein, 5.7 g/dL; albumin, 2.7 g/dL; total bilirubin, 0.4 mg/dL; aspartate aminotransferase, 134 U/L; alanine aminotransferase, 137 U/L; alkaline phosphatase, 203 U/L; gamma-glutamyl transferase, 18 U/L; and amylase, 68 U/L. C-reactive protein level was 24.37 mg/dL (normal range <0.3 mg/dL) and procalcitonin level was 7.17 ng/mL (normal range <0.05 ng/mL). Serologic tests for tumor markers were as follows: alpha-fetoprotein, 1.2 ng/mL (normal range, 0-20); CA 19-9, 46.8 U/mL (0-37).
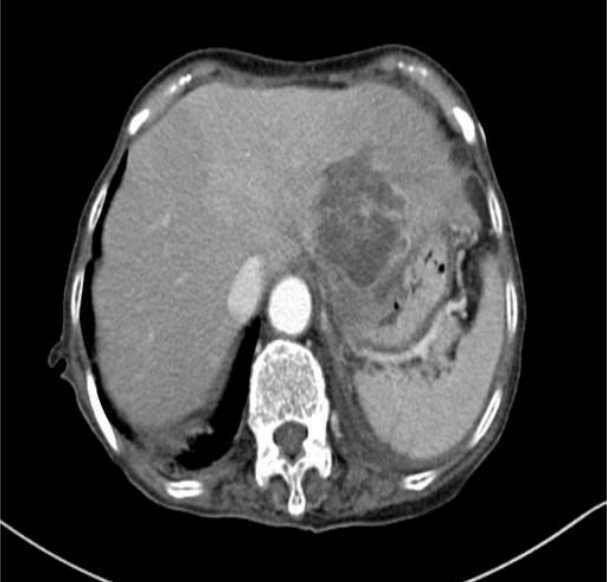
A computed tomography (CT) scan of the abdomen showed an approximately 5 cm sized lobulated hypodense lesion with internal septa and marginal enhancement in the left lateral section (Fig. 1). An early liver abscess was initially suspected; however, the coexistence of cholangiocarcinoma could not be completely ruled out. Initially, ultrasonography-guided aspiration or drainage was not performed due to the lesionŌĆÖs immature characteristics. An ultrasonography-guided biopsy was performed after 9 days of hospitalization due to the possibility of combined malignancy. Microscopic examination showed no malignant cells. Pus culture revealed Klebsiella pneumonia. After treatment with intravenous antibiotics for 14 days, fever subsided, and WBC count decreased to 8,910/╬╝L. Serum amylase was slightly increased (143 U/L). Blood cultures were sterile.
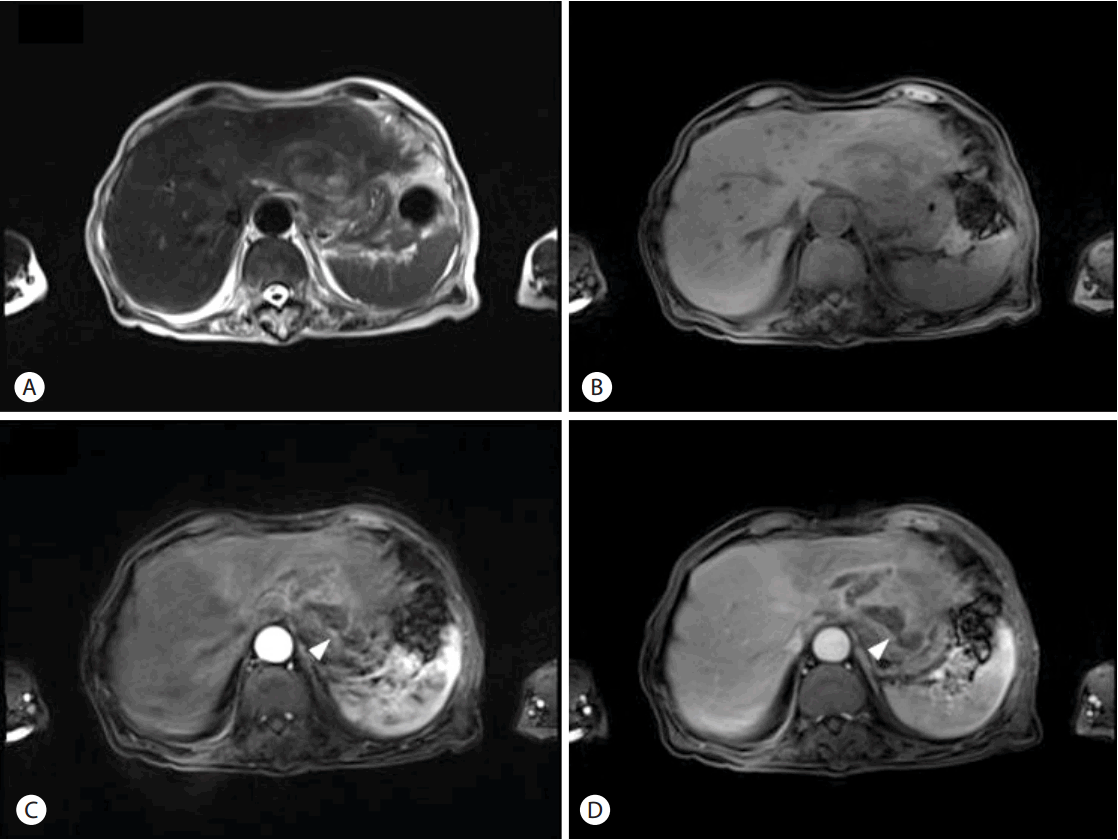
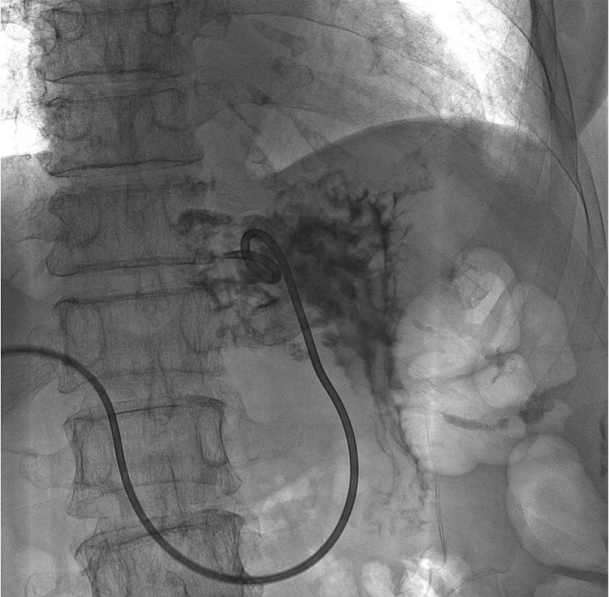
Gadoxetate-disodium-enhanced magnetic resonance imaging (Fig. 2) 17 days after hospitalization showed low T1 and high T2 signal intensities and rim-like enhancement in the left lateral section. The anterior wall of the stomach was in close proximity to the liver abscess, and communication between the gastric lumen and the liver was found (white arrowhead). Because magnetic resonance imaging showed a more mature appearance compared to the initial CT scan, ultrasonography-guided percutaneous drainage of the liver abscess was performed. After injection of contrast media through a pigtail catheter, the fistulous tract between the liver and stomach was visualized (Fig. 3).
Resection of the involved portion of the stomach and the lateral section of the liver was performed. Microscopic examination of the resected liver revealed abscess formation without malignancy. Biopsy of the resected stomach revealed inflammatory changes throughout the gastric wall. The patient was discharged from the hospital 2 weeks after surgery.
Liver abscesses may rupture into adjacent pleural, pericardial, and peritoneal cavities [1] however, fistulization into the gastrointestinal tract is rare [2-7]. To date, there have been only a handful of case reports of a rare complication following liver abscess (Table 1). In previous reports, hepatogastric fistula presented with melena [2], bilious vomiting [5], drainage of food particles from the catheter [3], and/or a sudden decrease in the size of hepatic lesions [7]. However, specific symptoms or signs were not present in our patient.
Diagnosis of hepatogastric fistula is generally based on imaging and endoscopic findings. On CT, an abscess cavity with an internal air pocket in continuity with the gastric lumen provides a clue to the presence of fistula. Fistulous openings in the base of the ulcer may be seen on esophagogastroduodenoscopy [4]. In our case, direct communication between the liver abscess and the stomach was detected on imaging. Owing to the absence of specific manifestations, the diagnosis of hepatogastric fistula was made accidentally on magnetic resonance imaging and tubography during insertion of a drainage catheter in this patient.
In our case, the drainage procedure was deferred due to the initial immature features of the abscess cavity, but resulted in spontaneous fistulization into the stomach. This implies that proper drainage of pyogenic liver abscesses should be recommended to prevent spontaneous abscess rupture into adjacent structures. Early drainage of liver abscess to prevent such complication was also highlighted on previous reports [7,8]. Definitive guidelines for the management of hepatogastric fistula have not to be identified; in general, surgical treatment is recommended as a definitive therapy. Conservative management including antibiotics and proton pump inhibitors has also led to spontaneous closure of fistulas [2-4,6]. We have managed patients surgically for the curative treatment of liver abscess and fistula.
In summary, hepatogastric fistula is a rare complication of liver abscess. The possibility of hepatogastric fistula should be kept in mind in patients with liver abscess for which drainage is delayed, even when there are no specific symptoms. Early drainage is needed to avoid spontaneous rupture into the gastrointestinal tract.
Figure┬Ā1.
Arterial-phase CT revealed an approximately 5-cm-sized lobulated hypodense lesion with peripheral rim enhancement in the left hepatic lobe. CT, computed tomography.
Figure┬Ā2.
Magnetic resonance imaging of the liver showed a multiseptated lesion with peripheral high signal intensity on (A) T2-weighted imaging and low signal intensity on (B) unenhanced T1-weighted imaging. The peripheral portion was enhanced on (C) arterial- and (D) portal-phase MRI. The central portion was more ripened compared to that of the initial CT scan. This hepatic lesion appeared to be in direct continuity with the stomach lumen (white arrowhead). MRI, magnetic resonance imaging; CT, computed tomography.
Figure┬Ā3.
Tubography revealed contrast media leakage via a fistulous tract between the liver and stomach.
Table┬Ā1.
Hepatogastric fistula as a complication of liver abscess. Review of the literature
| Author (Year) | Age/Sex | Cause | Location of liver abscess | Size | Symptoms or signs of hepatogastric fistula | Diagnosis | Treatment | Prognosis |
|---|---|---|---|---|---|---|---|---|
| De Lima et al. [9] (1989) | 36/F | Amoebic | Left lobe | Not described | Not described | UGIS | Medical | Improved |
| P├╝sp├Čk et al. [10] (2000) | 41/M | Amoebic | Left lobe | 8 cm | Anemia | EGD | Medical | Improved |
| Tomiyama et al. [8] (2007) | 77/F | Pyogenic (K. pneumoniae) | Left lobe | 13 cm | Increase in size of cystic liver mass | CT, EGD | Surgical | Improved |
| Monge-Fresse et al. [7] (2007) | 44/M | Pyogenic | Left lobe | 5.5 cm | Decrease in size of liver abscess | CT | Medical | Improved |
| Hsu et al. [6] (2009) | 86/M | Pyogenic (K. pneumoniae) | Left lobe | 6.5 cm | Dramatically decrease in drain amount from pigtail | Tubography | Medical | Improved |
| Kumar et al. [3] (2011) | 38/M | Amoebic | Bilobar | Not described | Food particles in the catheter | CT | Medical | Improved |
| Takahashi et al. [11] (2011) | 26/M | Amoebic | Left lobe | 8 cm | Not described | EGD | Medical | Improved |
| Singh et al. [12] (2013) | 35/M | Amoebic | Left lobe | 15 cm | Initial presentation | CT | Surgical | Improved |
| Kim et al. [2] (2013) | 64/F | Pyogenic | Left lobe | 3 cm | Melena | EGD | Medical | Improved |
| Gandham et al. [5] (2014) | 40/M | Pyogenic (K. pneumoniae) | Bilobar | Not described | Intermittent large-quantity billous vomiting | EGD | Medical, endoscopic stenting | Improved |
| Pawar et al. [4] (2015) | 47/M | Amoebic | Bilobar | 13.7 cm | Melena | EGD | Medical | Improved |
REFERENCES
1. Chan KS, Chen CM, Cheng KC, Hou CC, Lin HJ, Yu WL. Pyogenic liver abscess: a retrospective analysis of 107 patients during a 3-year period. Jpn J Infect Dis 2005;58:366-368.


2. Kim ES, Lee SY, Yeom JO, Cho YS. Education and Imaging. Hepatobiliary and pancreatic: Liver abscess complicated by a hepatogastric fistula. J Gastroenterol Hepatol 2013;28:206.


3. Kumar R, Sundar S, Sharma P, Sarin SK. Conservative management of liver abscess complicated by hepatogastric fistula. Dig Liver Dis 2011;43:752-753.


4. Pawar SV, Zanwar VG, Gambhire PA, Mohite AR, Choksey AS, Rathi PM, et al. Unusual complication of amebic liver abscess: Hepatogastric fistula. World J Gastrointest Endosc 2015;7:916-919.



5. Gandham VS, Pottakkat B, Panicker LC, Hari RV. Hepatogastric fistula: a rare complication of pyogenic liver abscess. BMJ Case Rep 2014;2014.



6. Hsu CY, Liu JH, Su CW. Hepatogastric fistula in one patient with liver abscess. Gastroenterology 2009;137:e7-8.

7. Monge-Fresse AS, Siriez JY, Bricaire F. Spontaneous fistulisation of a liver abscess into the stomach. Neth J Med 2007;65:456-457.

8. Tomiyama Y, Ohmoto K, Yoshioka N, Kawase T, Shibata N, Yoshida K, et al. [Pyogenic liver abscess complicated by gastric fistula and bacterial meningitis]. Nihon Shokakibyo Gakkai Zasshi 2007;104:1512-1518.

9. De Lima E, Mayoral LG, Falabella F, Rengifo A. Gastric perforation of amoebic liver abscess: report of a case. Trans R Soc Trop Med Hyg 1989;83:79-80.


10. P├╝sp├Čk A, Kiener HP, Susani M, M├╝ller C. Gastric perforation of a left lobe amoebic liver abscess. Eur J Gastroenterol Hepatol 2000;12:961-962.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




