Clinical practice guidelines and real-life practice on hepatocellular carcinoma: A the Hong Kong perspective
Article information
Abstract
Hepatocellular carcinoma (HCC) is a major public health burden in Hong Kong, and chronic hepatitis B is the most common HCC etiology in our region. With the high case load, extensive local expertise on HCC has been accumulated. This article summarized local guidelines and real-life practice on HCC management in Hong Kong. For HCC surveillance, liver ultrasound and serum alpha-fetoprotein for periodic screening is recommended in viral hepatitis or cirrhotic patients, and this is adhered to in clinical practice. HCC diagnosis is not covered in local guidelines, yet our practice is in-line with regional guidelines, where diagnosis is usually achieved by cross-sectional imaging and without the need for histology. Our guidelines recommend using the Hong Kong Liver Cancer Staging for pre-treatment staging, yet we routinely use other widely-adopted systems such as the Barcelona Clinic Liver Cancer Staging and the Tumor-Node-Metastasis Staging as well. Our local guidelines have provided clear treatment algorithms for the whole range of HCC therapies, including resection, ablation, transplant, transarterial chemoembolization, transarterial radioembolization, stereotactic body radiation therapy, targeted therapy, and immunotherapy. Real-life treatment choices are largely in line with the guidelines, although treatment protocols are individualized, and availability of specific therapies can vary between centers. Overall, HCC guidelines in Hong Kong are tailored based on local expertise and our unique patient population. The guidelines are up-to-date and provide practical pathways to assist our routine practice. Regular updates of local guidelines are warranted to account for the rapidly evolving paradigm of HCC management.
INTRODUCTION
Liver cancer is the fifth most common cancer and the third leading cause of cancer death in Hong Kong [1]. Among primary liver cancers, hepatocellular carcinoma (HCC) is the major type which accounts for 90% of cases. From 1992 to 2006, around 80% of HCC in Hong Kong were attributable to chronic hepatitis B (CHB), and only 6.3% were attributable to chronic hepatitis C (CHC) [2]. This contrasts with the key HCC risk factors of CHC, non-alcoholic steatohepatitis (NASH), and alcoholic liver disease in Western countries [2].
CHB-related HCC patients generally have better preserved liver function than HCC patients with other liver diseases, as hepatitis B virus (HBV) is directly oncogenic and can induce HCC in non-cirrhotic patients [3]. Furthermore, given the high case load and experience in HCC management, experts in Hong Kong generally propose more aggressive treatment approaches than that recommended in international guidelines.
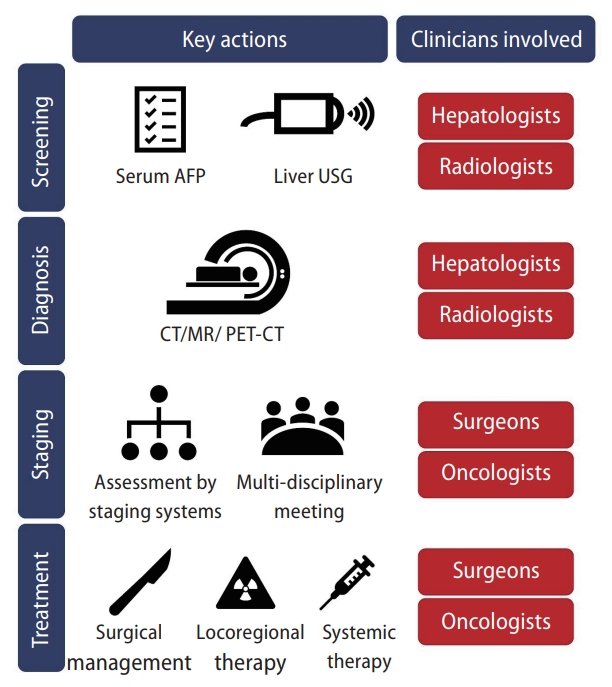
This article will discuss both the local guidelines and real-life practice on HCC care in Hong Kong, covering the areas of screening, diagnosis, staging and treatment (Table 1). The management framework of HCC in Hong Kong is depicted in Figure 1. The key guidelines covered include (1) Recommendations on Prevention and Screening for Liver Cancer for Health Professionals (Published in 2018) [4]; (2) The Hong Kong Liver Cancer (HKLC) Staging system (Published in 2014) [3]; (3) The Hong Kong Consensus Statement on the Management of Hepatocellular Carcinoma (Hong Kong Consensus) (Published in 2022) [5]; and (4) Systemic Treatment of Advanced Unresectable Hepatocellular Carcinoma after First-Line Therapy: Expert Recommendations from Hong Kong, Singapore, and Taiwan (Published in 2022) [6].
SURVEILLANCE
Clinical guidelines
The Hong Kong Cancer Expert Working Group on Cancer Prevention and Screening published recommendations on HCC screening in 2018 [4]. The guidelines recommend the combined use of liver ultrasound (USG) and serum alpha-fetoprotein (AFP) for HCC screening, whereas computed tomography (CT) and magnetic resonance (MR) scans are recommended in patients with suboptimal USG assessment. CHB, CHC and cirrhotic patients are recommended to receive period screening (every 6–12 months), depending on age, family history and other clinical parameters.
Real-life practice
Patients with chronic liver diseases are usually followed-up by hepatologists in Hong Kong, and eligible patients are advised for screening at 6-month intervals. Local data has demonstrated that screening by USG and/or AFP is associated with HCC detection at earlier stages and improved survival [7].
The availability of CT and MR scans is increasing in our locality, and patients with suboptimal USG assessment (such as in obesity, ascites or high-lying liver) are referred for CT or MR. The use of artificial intelligence on CT scans [8] and advanced MR imaging metrics [9] have also been explored as surveillance options in exploratory studies in Hong Kong.
DIAGNOSIS
Clinical guidelines
No local guidelines have focused on HCC diagnosis in Hong Kong, and Asia-Pacific regional guidelines are frequently referenced to by clinicians. The Asia-Pacific guidelines recommend triphasic CT or gadoxetic acid-enhanced MR for diagnosing HCC [10]. Imaging findings are classified by the Liver Reporting & Data System, with major diagnostic features defined by arterial phase enhancement, non-peripheral washout and enhancing capsule [11]. Histology is not required for diagnosis if imaging features are diagnostic [10].
Real-life practice
Imaging is the predominant modality for HCC diagnosis in Hong Kong, and histology is rarely required. Triphasic CT is available in most tertiary hospitals in our locality, and it is the most frequently utilized test for HCC diagnosis [3]. Nonetheless, gadoxetic acid-enhanced MR has risen in popularity in recent years for its higher sensitivity and better lesion delination [12].
Dual-tracer positron emission tomography-CT (PET-CT) with 11C-Acetate and 18F-fluorodeoxyglucose is another increasingly popular diagnostic test for HCC in our locality. Dual-tracer PET-CT is usually used as a confirmatory test in patients with indeterminate findings on CT or MR. Local data has demonstrated that dual-tracer PET-CT is significantly less affected by cirrhotic changes than contrast CT for HCC diagnosis [13]. Furthermore, dual-tracer PET-CT has the additional benefit of detecting metastatic foci, aiding the disease staging process [14]. A cost-effectiveness study using local data demonstrated that dual-tracer PET-CT is cost-effective for assessment in patients with AFP ≥400 ng/mL or with bilobar disease [15].
STAGING
Clinical guidelines
The HKLC Staging System is recommended as the staging system of choice in the Hong Kong Consensus Guidelines [5]. The HKLC Staging was developed in 2014 with data from 3,856 HCC patients in Hong Kong. The rationale for developing the HKLC Staging was to account for the high HBV prevalence and generally more aggressive treatment approach in Hong Kong [3]. The prognostic factors in the HKLC Staging include Eastern Cooperative Oncology Group status, Child-Pugh score, presence of extravascular invasion or metastasis, tumor size and number of tumor nodules (Fig. 2) [3].
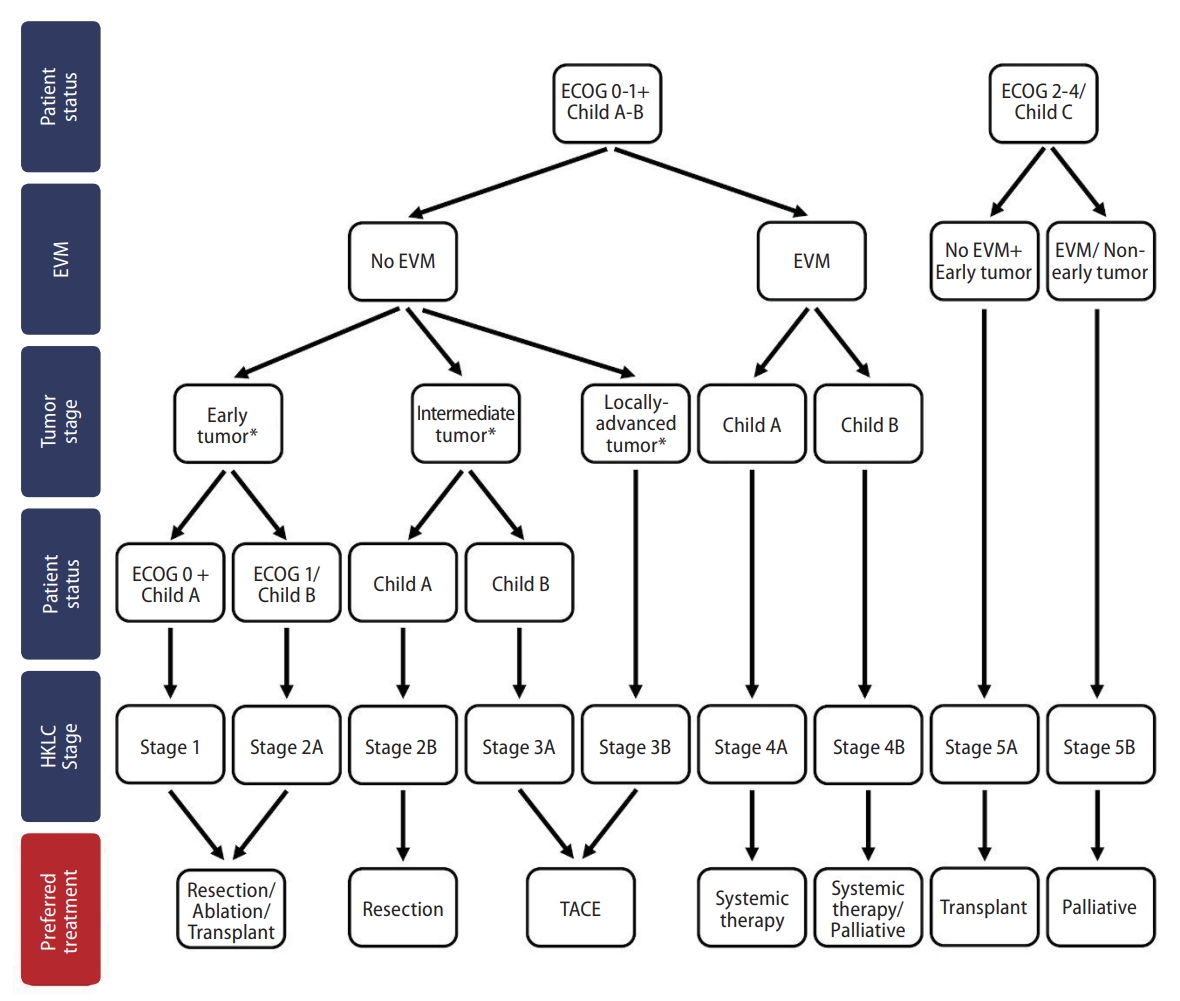
Staging and preferred treatment in the Hong Kong Liver Cancer Staging System. ECOG, Eastern Cooperative Oncology Group; EVM, extrahepatic vascular invasion or metastasis; TACE, transarterial chemoembolization; HKLC, Hong Kong Liver Cancer. *Early tumor: ≤5 cm+≤3 tumor nodules+No intrahepatic venous invasion; Intermediate tumor: ≤5 cm+>3 tumor nodules/Intrahepatic venous invasion OR >5 cm+≤3 tumor nodules+No intrahepatic venous invasion; Locally-advanced tumor: ≤5 cm+>3 tumor nodules+intrahepatic venous invasion OR >5 cm+>3 tumor nodules+intrahepatic venous invasion OR diffuse tumor.
Real-life practice
After the diagnosis of HCC in Hong Kong, patients are referred from hepatologists to specialized HCC clinics, where they receive joint care by surgeons and oncologists to individualize their next step of treatment. Hepatologists will regularly review the patients and advise on management of the primary liver disease (e.g., antiviral use) and on cirrhotic complications (e.g., diuretic titration, variceal screening). However, the anti-cancer therapy decisions including planning for surgical therapy, TACE, or systemic therapy are primarily handled by surgeons and oncologists.
When comparing the HKLC Staging with the widely adopted Barcelona Clinic Liver Cancer (BCLC) Staging, HKLC more clearly delineates intermediate or advanced stage patients into smaller subgroups, enabling different treatment strategies for this heterogenous patient group [3]. Models have shown HKLC to have better prognostic performance over other staging systems in Hong Kong [3]. Nonetheless, traditional staging systems such as the BCLC Staging and the Tumor-Node-Metastasis System remain frequently used in clinical practice and research in Hong Kong, possibly due to historical reasons and for ease of communication [16-20].
CURATIVE TREATMENT – RESECTION AND ABLATION
Clinical guidelines
The Hong Kong Consensus recommends liver resection as the first-line treatment for HCC with solitary lesion or with several lesions limited to segment(s) with resectable potential, given satisfactory liver function reserve. For patients with inadequate future liver remnant, portal vein embolization (PVE) or associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) are both suitable to enhance resectability.
Portal vein invasion, extrahepatic metastasis and bilobar disease are traditionally listed as contraindications for hepatectomy in Western guidelines, whereas the Hong Kong Consensus recommends consideration of hepatectomy in highly-selected patients. For patients with intrahepatic portal vein or hepatic vein branch invasion, resection can be considered in specialized centers. For limited extrahepatic metastasis and bilobar disease, the predominant HCC lesion can be resected, while the extrahepatic metastasis or contralateral liver lobe nodules can be managed by resection or ablation [5].
Local ablation is recommended as an alternative to resection in Child-Pugh A/B patients with HCC <3 cm. The options of radiofrequency ablation (RFA), microwave ablation (MWA), high-intensity focused ultrasound (HIFU), and cryoablation are all suitable, whereas percutaneous ethanol injection is less preferred due to lower complete ablation rates and higher recurrence rates [5].
Real-life practice
In Hong Kong, liver resection is the main treatment option for early-stage HCC. The pre-hepatectomy workup involves indocyanine green retention testing and CT volumetry assessment [21]. For patients with inadequate future liver remnant, ALPPS or PVE may be offered, although the availability of ALPPS is limited and is only performed in specialized centers. A local study conducted in 2021 reported significantly higher resection rates for ALPPS when compared with PVE (97.8% vs. 67.7%), although the two methods conferred comparable short-term and long-term mortality [21].
Hepatectomy in patients with portal vein thrombosis has been performed for over three decades in Hong Kong. The median disease-free survival ranged from 1.5 to 4.2 months, and overall survival ranged from 8.6 to 10.9 months [22]. These recurrence and mortality rates are in line with data from other regions [23].
Combined resection and intraoperative RFA for multifocal HCC have also been performed in Hong Kong. Combined resection and RFA, when compared with resection alone, led to fewer major resections, less blood loss, shorter operation time and shorter hospital stay, with no differences in recurrence and overall survival [24]. Local data is also available for patients with resectable HCC with lung metastasis, and successful resection of lung metastasis after hepatectomy is associated with significant improvement in survival [25].
A randomized trial in Hong Kong performed head-to-head comparison between RFA and resection for early-stage HCC. RFA led to shorter treatment duration, shorter hospital stay, and lower blood loss, although the recurrence rate and 10-year overall survival were comparable [26]. RFA is feasible and frequently considered in Hong Kong for patients with high surgical risk. Other ablation options such as MWA and cryoablation are less frequently performed locally.
CURATIVE TREATMENT – LIVER TRANSPLANT
Clinical guidelines
Liver transplantation is recommended by the Hong Kong Consensus in patients with poor liver function, as it can treat both HCC and cirrhosis [5]. The guidelines recommend patient selection for transplant by the University of California San Francisco criteria – Solitary tumor <6.5 cm; or <3 tumor nodules with largest lesion <4.5 cm with total tumor diameter <8.0 cm [27].
For patients waitlisted for transplant, options of bridging therapy include local ablation, transarterial chemoembolization (TACE), transarterial radioembolization (TARE) and stereotactic body radiation therapy (SBRT) [5].
Real-life practice
Our hospital (Queen Mary Hospital, Hong Kong) is the only designated liver transplant center in Hong Kong, where both deceased-donor liver transplants (DDLTs) and living-donor liver transplants (LDLTs) are performed. All patients waitlisted for transplant are entered into a central transplant registry, and cadaveric donor liver grafts are allocated based on the Model for End-stage Liver Disease score, with adjustment for HCC status. However, cadaveric liver donation is uncommon in Hong Kong, possibly due to societal and cultural reasons [28]. In the past decade, the annual number of liver donations from deceased donors has remained less than 40 [29].
To tackle the scarcity of deceased donor liver grafts, LDLT using extended right lobe grafts for adults has been pioneered in Hong Kong since 1996 [30], and the technique has gained popularity in Asia. From 1996 to 2018, over 1,400 LDLTs have been performed in Hong Kong, with 16.5% of LDLTs performed for HCC [31].
A local prospective study has compared LDLT against hepatectomy for HCC, where LDLT demonstrated a 1.5-fold higher 10-year overall survival (83% vs. 56% respectively) with 2-fold higher 10-year disease-free survival (81% vs. 40% respectively) [32]. Local data has also reported comparable 5-year oncological outcomes and survival when comparing LDLT and DDLT for HCC [33]. LDLT is the predominant type of transplant surgery in Hong Kong, and it is an ideal HCC treatment in our locality.
All the recommended bridging therapy options, including local ablation (RFA or HIFU), TACE, TARE and SBRT are performed in Hong Kong [34]. In particular, TACE and SBRT are the most commonly performed. A recent local study demonstrated bridging SBRT was associated with a significantly higher 1-year tumor control when compared with TACE or HIFU, supporting SBRT as the top choice for bridging [35].
CURATIVE TREATMENT – POST-TREATMENT SURVEILLANCE AND MANAGEMENT
Clinical guidelines
No local guidelines in Hong Kong have covered post-treatment surveillance and management. Nonetheless, the US National Comprehensive Cancer Network recommendations are frequently referenced, where patients receive CT/MR scans plus serum AFP testing every 3–6 months for 2 years after treatment, followed by surveillance at 6–12 month intervals thereafter [36].
Real-life practice
Surveillance after curative HCC therapy is overseen by surgeons in Hong Kong [37,38]. Close surveillance by CT/MR with AFP is generally adhered to, enabling early detection and management of recurrence. The use of dual-tracer PET-CT as surveillance has also been explored in post-transplant patients in a local study. Notably, dual-tracer PET-CT had increased sensitivity of recurrence detection by 12%, and led to change in management in one-third of post-transplant patients [38].
As the majority of HCC in Hong Kong are CHB-related, antivirals are routinely used to reduce the recurrence risk after curative therapy for HCC. Despite close surveillance and antiviral therapy, local data has shown HCC recurrence rates of over 70% in hepatectomy and over 20% in liver transplant respectively, which are compatible with international data [37,38]. In patients with recurrence, treatment protocols are generally similar to that in patients with newly diagnosed HCC.
NON-CURATIVE TREATMENT – LOCOREGIONAL THERAPY
Clinical guidelines
TACE is recommended in unresectable HCC with no vascular invasion or extrahepatic spread and with satisfactory liver function. While patients with no vascular invasion are ideal candidates, segmental portal vein thrombosis and venous invasion are not absolute contraindications. TACE with drugeluting beads (DEB-TACE) is a possible alternative if conventional TACE fails to achieve a tumor response [5].
Besides TACE, SBRT and TARE are other options for locoregional therapy for Child-Pugh A patients, especially for larger tumors (>5 cm). In particular, the versatility of SBRT is highlighted in that SBRT may be used for bridging to transplant, for downstaging HCC for resection, and to be used in combination with TACE [5].
Real-life practice
TACE is the first-line therapy for unresectable intermediate stage or locally-advanced HCC in Hong Kong. DEB-TACE is also available in some tertiary centers. A shared-care approach between surgeons and oncologists enable smooth transitioning of TACE-refractory patients to systemic therapy [5].
The use of SBRT for unresectable HCC, either alone or in combination with TACE, is increasing in Hong Kong [39]. Local experience has demonstrated that SBRT+TACE, when compared with TACE alone, led to significantly improved radiological disease control (98.0% vs. 56.7%), 1-year overall survival (67.2% vs. 36.5%), and 3-year overall survival (43.9% vs. 13.3%) [40].
TARE is less frequently performed than TACE in Hong Kong. Limited local data has shown that TARE is safe and has similar 1-year survival when compared with TACE [41,42].
NON-CURATIVE TREATMENT – SYSTEMIC THERAPY
Clinical guidelines
Systemic therapy is the last therapeutic option in patients with metastatic disease or in advanced HCC patients who have failed locoregional therapy.
In the 2022 local guidelines, the combination of Atezo-lizumab+Bevacizumab is recommended as first-line systemic treatment, although this recommendation is restricted to patients with good liver function (Child-Pugh A). Nivolumab monotherapy is an alternative first-line systemic treatment for Child-Pugh B patients, and may be considered for patients with poorly-controlled hypertension or high bleeding risks. Tyrosine kinase inhibitor (TKIs) (Lenvatinib or Sorafenib) are also suitable as first-line treatment if the patient prefers oral treatment or have contraindications for immunotherapy [5,6].
For patients who progress after first-line systemic therapy, second-line targeted therapy including Regorafenib, Cabozantinib or Ramucirumab are suitable options [5,6]. Immunotherapy may be considered as second-line treatment for patients who failed first-line TKIs [6].
Real-life practice
A range of international drug trials are ongoing in the academic centers in Hong Kong, and advanced HCC patients may be able to receive novel agents in drug trials [43,44]. Outside of the context of clinical trials, the whole range of guidelinerecommended targeted therapy and immunotherapy options are available locally. Nonetheless, treatment protocols are usually individualized due to patient preferences and tolerability. In Hong Kong, the use of targeted therapy and immunotherapy as approved therapies have to be paid out-of-pocket by patients, although subsidies may be available for some options from the government and non-government organizations. Cost concern is hence another important factor that impact drug choices for our HCC patients.
In recent years, the use of immunotherapy (Nivolumab monotherapy or Nivolumab+Ipilimumab) or immunotherapy combined with anti-angiogenic agents (e.g., Atezolizumab+Bevacizumab) as first-line systemic therapy is increasingly advocated in our locality due to data from the landmark IMbrave150, CheckMate459 and Checkmate040 trials respectively [45-47]. Bleeding risks and cost are potential reasons in favour of using Nivolumab-containing regimens over Atezolizumab+Bevacizumab.
Poor liver reserve in some patients preclude them from receiving immunotherapy, and patients’ preferences for oral drugs are also frequently encountered in our locality [48]. These factors lead to first-line TKIs to still have a prominent role for systemic therapy in Hong Kong. While Sorafenib and Lenvatinib are both recommended as first-line TKIs, Lenvatinib is generally preferred over Sorafenib in Hong Kong due to the superior disease control and overall response rate of Lenvatinib in the REFLECT trial [49].
As per the guidelines, second-line use of immunotherapy or targeted therapy is practiced in Hong Kong [48]. However when patients progress beyond second-line treatment, no clear instructions are available in the local guidelines, and treatment is based on expert-opinion. Exciting yet limited data has emerged in Hong Kong for treatment beyond second-line. For example, the combination of Nivolumab/Pembrolizumab+Ipilimumab has been used in patients who have failed prior immune checkpoint inhibitor therapy, where 1-year overall survival of 42.4% was achieved, with 12.0% of patients achieving complete response [17]. The novel combination of Cabozantinib (TKI) with immunotherapy has also been reported in 15 patients, achieving a promising 1-year survival of 71.5% [50].
DISCUSSION
In the past few decades, researchers from Hong Kong have produced high-impact studies that have shifted the paradigm of HCC management. With support from local data, multidisciplinary teams of practicing clinicians have developed local HCC guidelines that are up-to-date and practical.
In Hong Kong, deviation of real-life practice from HCC guidelines may represent efforts to improve HCC care based on novel data and expert experience. For example, the first-line use of dual-tracer PET-CT and the “off-label” use of immunotherapy for treatment-resistant HCC are exciting developments that may be increasingly adopted and may be incorporated in future versions of the guidelines. Real-life practice may also vary due to resource constraints and availability of expertise. Nonetheless, Hong Kong is unique in its relatively small geographical size (around 1,100 km2 only), and referral of patients to specialized tertiary centers is easily achievable and frequently performed [51].
Despite the strengths of the HCC guidelines in Hong Kong, several areas can be improved on. First, the current guidelines have not provided recommendations on HCC diagnosis. As Hong Kong has an increasing availability of advanced imaging services [9,14,20], local guidelines on diagnostic pathways will be beneficial to enable earlier and more accurate diagnosis of HCC.
Second, post-treatment surveillance and risk reduction are important areas that should be discussed in the guidelines as well. The use of antivirals for CHB is routinely used to reduce HCC recurrence in Hong Kong. Nonetheless, local data has highlighted other risk factors for HCC in CHB patients such as fibrotic burden and metabolic risk factors [52-54]. In fact, a local study on a mixed cohort of HCC patients highlighted hepatic steatosis as a key risk factor for HCC recurrence [20]. Guidelines are warranted to guide the post-treatment follow-up plan for HCC, with potential need to include metabolic screening as a measure to reduce recurrence.
Finally, supportive care for HCC is an area that has not been covered in the local guidelines. Palliative care in hepatology is a topic that is gaining increasing attention in recent years [55]. In particular, palliative care in HCC is unique given the uncertain disease course, risk of rapid progression, and the need to care for both malignancy-related and cirrhosis-related symptoms [56]. Guidance for symptomatic care may improve the quality-of-life for advanced HCC patients that have failed other therapies.
Overall, the HCC guidelines in Hong Kong are practical and up-to-date. The guidelines are tailored based on our unique population and local expertise, and are able to assist our daily practice. They are mostly adhered to in the current clinical practice in Hong Kong. Nonetheless, the field of HCC management is rapidly evolving, and regular updates of our guidelines will be required to incorporate the novel data that is consistently emerging from local and international studies.
Notes
Authors’ contribution
RWHH was involved in data interpretation and drafting of the manuscript. LYM, TTC, VHFL and WKS were involved in critical revision of the manuscript. MFY was involved in study concept, critical revision of the manuscript, and overall study supervision. All authors have seen and approved the final version of the manuscript.
Conflicts of Interest
MF Yuen is an advisory board member and/or received research funding from AbbVie, Arbutus Biopharma, Assembly Biosciences, Bristol Myer Squibb, Dicerna Pharmaceuticals, MF Yuen is an advisory board member and/or received reBiosciences, Bristol Myer Squibb, Dicerna Pharmaceuticals, GlaxoSmithKline, Gilead Sciences, Janssen, Merck Sharp and Dohme, Clear B Therapeutics, Springbank Pharmaceuticals; and received research funding from Arrowhead Pharmaceuticals, Fujirebio Incorporation and Sysmex Corporation. WK Seto received speaker’s fees from AstraZeneca and Mylan, is an advisory board member of CSL Behring, is an advisory board member and received speaker’s fees from AbbVie, and is an advisory board member, received speaker’s fees and researching funding from Gilead Sciences. The remaining authors have no conflict of interests.
Abbreviations
AFP
alpha-fetoprotein
ALPPS
associating liver partition and portal vein ligation for staged hepatectomy
BCLC Staging
Barcelona Clinic Liver Cancer Staging
CHB
chronic hepatitis B
CHC
chronic hepatitis C
CT
computed tomography
DDLT
deceased-donor liver transplant
DEB-TACE
TACE with drug-eluting beads
ECOG
Eastern Cooperative Oncology Group
HBV
hepatitis B virus
HCC
hepatocellular carcinoma
HIFU
high-intensity focused ultrasound
HKLC Staging System
Hong Kong Liver Cancer Staging System
ICG
indocyanine green
LDLT
living-donor liver transplant
LI-RADS
Liver Reporting & Data System
MELD
model for end-stage liver disease
MR
magnetic resonance
MWA
microwave ablation
NASH
non-alcoholic steatohepatitis
PET-CT
positron emission tomography-computed tomography
PVE
portal vein embolization
RFA
radiofrequency ablation
SBRT
stereotactic body radiation therapy
TACE
transarterial chemoembolization
TARE
transarterial radioembolization
TKI
tyrosine kinase inhibitor
TNM Staging
Tumor-Node-Metastasis Staging
UCSF
University of San Francisco
USG
ultrasound