Is liver biopsy essential to identifying the immune tolerant phase of chronic hepatitis B?
Article information
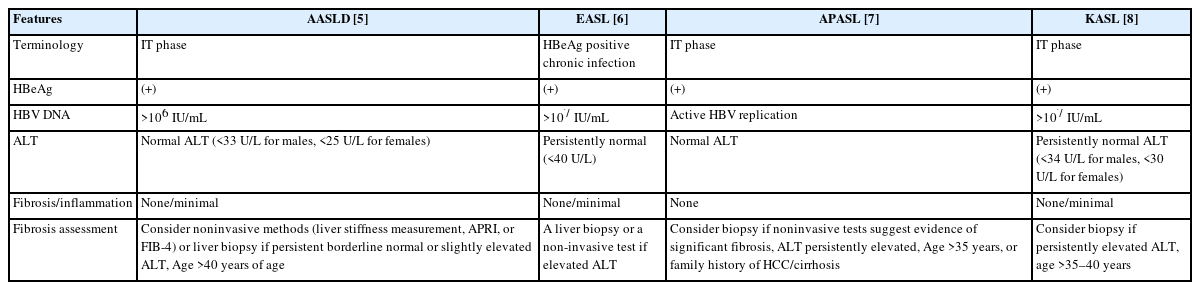
The natural history of chronic hepatitis B virus (HBV) infection is complex; it is characterized by different immune phases that may overlap [1]. The first phase is an immune-tolerant phase that is considered to be essentially benign in nature [2]. This phase is characterized by hepatitis B e antigen (HBeAg) positivity, very high levels of HBV DNA, and persistent normal alanine aminotransferase (ALT) levels [3]. However, the notion that the immune-tolerant phase is truly benign in nature has been challenged [4]. In addition, it is often difficult to accurately diagnose the true immune-tolerant phase [3]. The criteria and terminology used to define this phase also vary across international guidelines. The American Association for the Study of Liver Disease (AASLD) guidelines define immune-tolerant phase by HBeAg positivity, high serum HBV DNA levels (>106 IU/mL), and normal serum ALT level (<33 U/L for males and <25 U/L for females) [5]. The European Association for the Study of the Liver (EASL) guidelines no longer use the term ‘immune-tolerant phase’. They use ‘HBeAg-positive chronic infection,’ which they characterize as HBeAg positivity, high serum HBV DNA levels (>107 IU/mL), and normal ALT levels (<40 U/L for both sexes) [6]. The Asia Pacific Association for the Study of the Liver (APASL) guidelines provide similar criteria without specific HBV DNA or ALT cut-off values [7]. The Korean Association for the Study of the Liver (KASL) guidelines define the immune-tolerant phase by HBeAg positivity, high serum HBV DNA level (>107 IU/mL), and normal serum ALT level (<34 U/L for males, <30 U/L for females) (Table 1) [8].
HBV is a non-cytopathic virus that does not directly cause hepatocyte death [4]. Liver injury is considered immune mediated [4]. The hallmark of the immune-tolerant phase is minimal or absent liver injury [8]. Hence, there should be no necroinflammation or liver fibrosis on histologic exam in an immune-tolerant phase [9]. As a highly sensitive marker of liver injury, serum ALT level is often elevated when liver injury is present [10]. In the immune-tolerant phase, ALT level should not be elevated. However, the cutoff used to define ‘normal’ serum ALT levels differs among international guidelines (Table 1). ALT is also an imperfect marker of liver injury. Studies have shown that ALT is an inadequate marker to correctly identify the immune-tolerant phase [2,11]. Thus, whether liver biopsy is necessary to correctly identify the immune-tolerant phase remains unclear.
In this issue of the Clinical and Molecular Hepatology, Yoo et al. [12] studied 259 CHB patients who underwent liver biopsy with an immune-tolerant phase that was serologically identified. Surprisingly, they found that only 82 (31.7%) of these patients were in an immune-tolerant phase based on histologic findings. The authors used a serum HBV DNA level cutoff of >106 IU/mL and serum ALT level cutoff of <60 IU/L for both sexes to identify an immune-tolerant phase. Such criteria were less stringent than the AASLD or the EASL criteria. This might have contributed to the high proportion of patients in the “grey zone”, a territory that does not fall into typical CHB clinical phases. For patients who fall into this zone, the extent of liver damage and prognosis are uncertain. It is important to note that the ability to differentiate between immune-tolerant phase and other phases may be impacted by the inclusion of “grey zone” patients in the analysis. However, even when the more stringent AASLD or EASL criteria were used to analyze the same patients, only 31.7% or 34.0% of serologically immune-tolerant patients were in the same phase based on histological findings. This suggests that relying solely on the clinical definition may not accurately predict the severity of liver damage in CHB patients. Therefore, it is necessary to perform liver biopsy to accurately identify patients who require close monitoring or antiviral therapy [1].
In this cohort, fibrosis-4 index (FIB-4) was not as good indicator, as previously reported [2]. In patients with a low FIB-4 index (<1.45), 18% had advanced fibrosis (≥F3). The risk of liverrelated events such as hepatocellular carcinoma, liver cirrhosis, liver transplantation, and death was significantly lower for patients in a histologically immune-tolerant phase. These findings suggest that liver biopsy might be necessary to identify those who are in a true immune-tolerant phase and show a benign clinical course. Nevertheless, liver biopsy is an invasive procedure with potential complications. Performing liver biopsy for all patients who are presumed to be in an immune-tolerant phase is unrealistic. The authors thus suggested an age cut-off of 35 years or older as a criterion when considering liver biopsy. However, questions remain due to the invasiveness of liver biopsy. What if liver biopsy reveals a histologically immune-tolerant phase for those who are aged 35? Do they need repeat liver biopsy as they age to determine whether they are still in the histologically immune-tolerant phase? If repeat biopsy is necessary, how often should it be performed? From this perspective, there should be other criteria, beyond age, to define a true immune-tolerant phase, which should be essentially benign in nature. Novel biomarkers, which can now be measured using blood, might play a role in defining disease phases [13]. Some potential biomarkers that have been investigated include serum cytokines [14], specific T-cell subsets [15], microRNA [16], and hepatitis B core-related antigen (HBcrAg) [17,18]. For instance, patients in an immune-tolerant phase have significantly higher levels of HBcrAg than those who are in an immune clearance phase or an inactive carrier phase [19]. Although these biomarkers alone cannot definitively identify the immune-tolerant phase in CHB patients, they might be a useful tool for predicting the immune-tolerant phase when they are used in combination with other clinical and laboratory parameters. As performing repeat liver biopsy to verify an immune-tolerant phase is unrealistic in clinical practice, studies using new novel non-invasive biomarkers are eagerly awaited.
Yoo et al. [12] performed a retrospective study at academic institutions, and thus selection and/or indication bias might help explain the high incidence of necroinflammation or fibrosis in the immune-tolerant phase. Nevertheless, this study provides a strong and clear message. Patient should not be considered to be in a ‘genuine’ immune-tolerant phase without liver biopsy. Due to several limitations, liver biopsy cannot be recommended for all patients who are presumed to be in an immune-tolerant phase in clinical practice. However, this study revealed that, without such histologic information, patients presumed to be in an immune-tolerant phase must be carefully evaluated using a variety of factors.
Notes
Authors’ contributions
JHO drafted the manuscript. DHS reviewed and finalized the manuscript.
Conflicts of Interest
The authors have no conflicts to disclose.
Abbreviations
HBV
hepatitis B virus
HBeAg
hepatitis B e antigen
ALT
alanine aminotransferase
AASLD
American Association for the Study of Liver Disease
EASL
European Association for the Study of the Liver
APASL
Asia Pacific Association for the Study of the Liver
KASL
Korean Association for the Study of the Liver
FIB-4
fibrosis-4
HBcrAg
hepatitis B core-related antigen