| Clin Mol Hepatol > Volume 29(3); 2023 > Article |
|
ABSTRACT
Background/Aims
Methods
Results
ACKNOWLEDGMENTS
FOOTNOTES
SUPPLEMENTAL MATERIAL
Supplementary┬ĀTable┬Ā2.
Supplementary┬ĀTable┬Ā3.
Supplementary┬ĀFigure┬Ā1.
Supplementary┬ĀFigure┬Ā2.
Supplementary┬ĀFigure┬Ā3.
Supplementary┬ĀFigure┬Ā4.
Supplementary┬ĀFigure┬Ā5.
Supplementary┬ĀFigure┬Ā6.
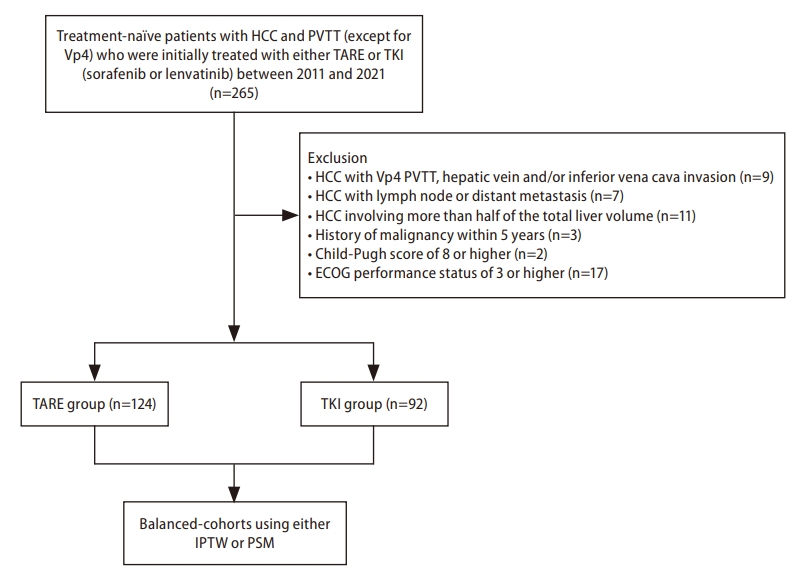
Figure┬Ā1.
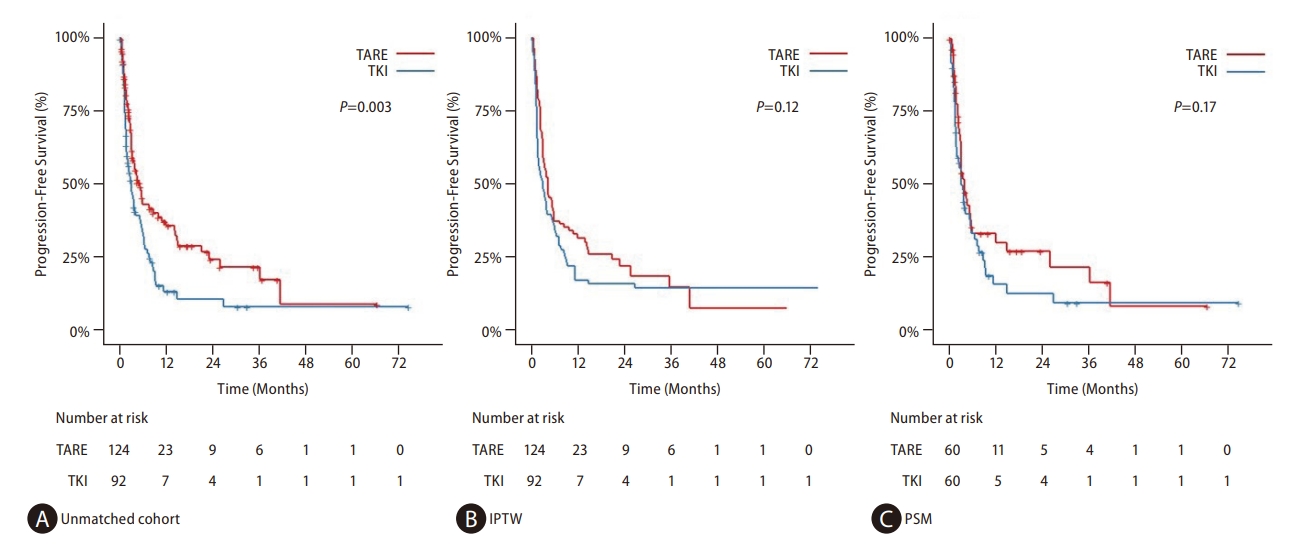
Figure┬Ā2.

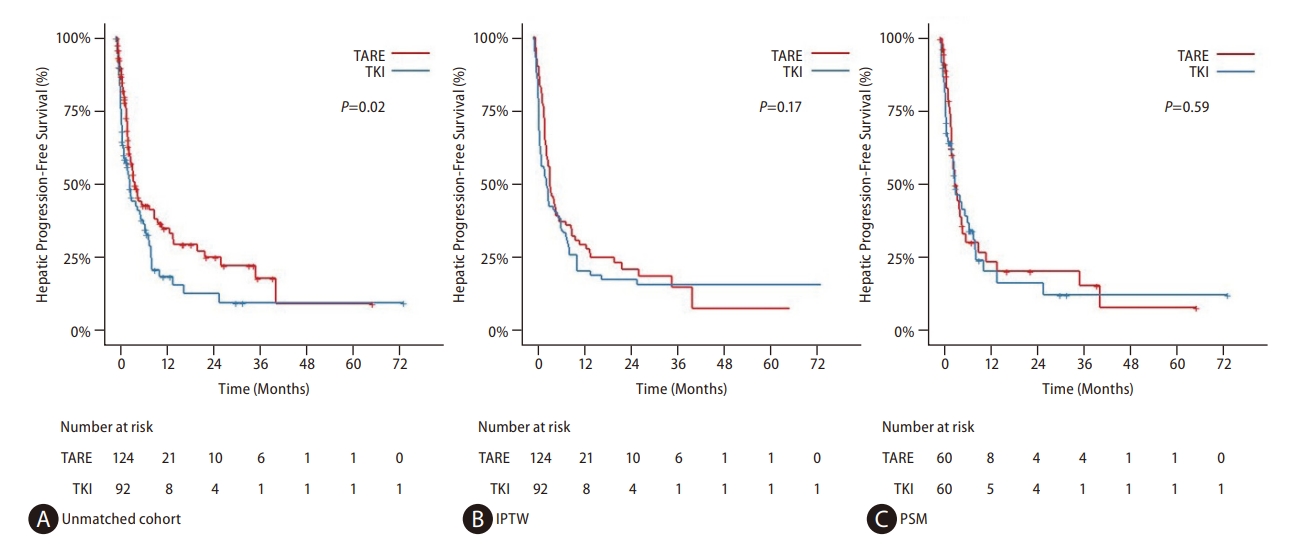
Figure┬Ā3.
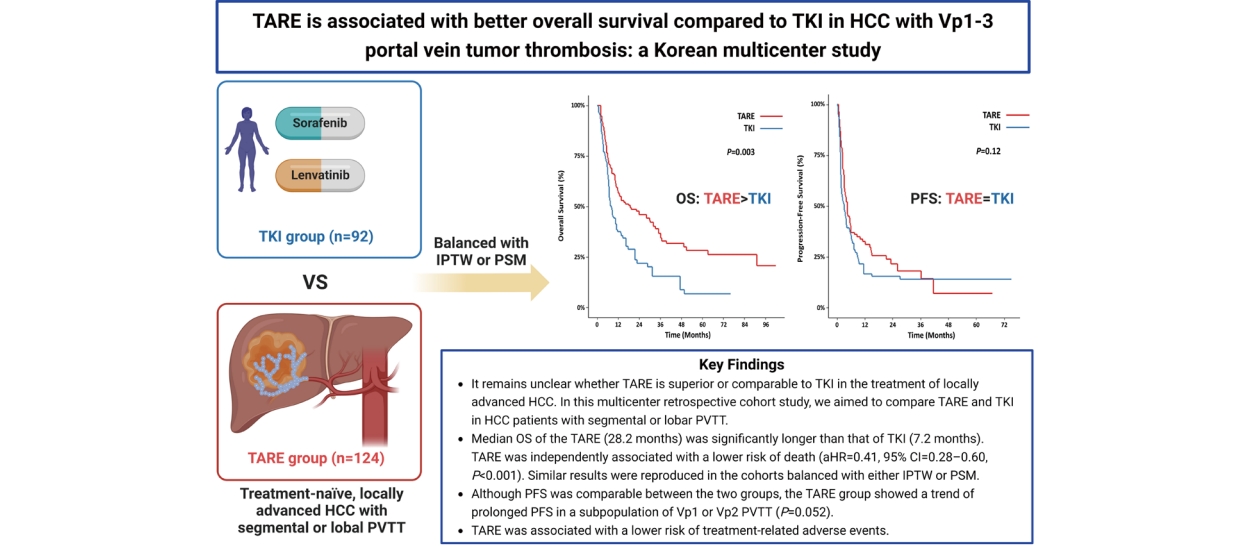
Figure┬Ā4.
Table┬Ā1.
Data are expressed as number (%) or median (interquartile range).
AFP, alpha fetoprotein; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ECOG, Eastern Cooperative Oncology Group; HBV, hepatitis B virus; HCV, hepatitis C virus; INR, international normalized ratio; IPTW, inverse probability of treatment weighting; PIVKA-II, protein induced by vitamin K absence II; PS, performance status; PSM, propensity score matching; PT, prothrombin time; PVTT, portal vein tumor thrombosis; TARE, transarterial radioembolization; TKI, tyrosine kinase inhibitor; WBC, white blood cell.
Table┬Ā2.
Data are expressed as median (interquartile range) or number (%).
CR, complete response; HPFS, hepatic progression-free survival; IPTW, inverse probability of treatment weighting; OS, overall survival; PD, progressive disease; PR, partial response; PFS, progression-free survival; PVTT, portal vein tumor thrombosis; SD, stable disease; TARE, transarterial radioembolization; TKI, tyrosine kinase inhibitor.
Table┬Ā3.
aHR, adjusted hazard ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HPFS, hepatic progression-free survival; OS, overall survival; progression-free survival; PFS, progression-free survival; PS, performance status; PT, prothrombin time; PVTT, portal vein tumor thrombosis; TARE, transarterial radioembolization; TKI, tyrosine kinase inhibitor.
Table┬Ā4.
CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HPFS, hepatic progression-free survival; IPTW, inverse probability of treatment weighting; OS, overall survival; PFS, progression-free survival; PS, performance status; PSM, propensity score matching; PT, prothrombin time; PVTT, portal vein tumor thrombosis; TARE, transarterial radioembolization; TKI, tyrosine kinase inhibitor.
Abbreviations
REFERENCES
- TOOLS
-
METRICS

- ORCID iDs
-
Yoon Jun Kim

https://orcid.org/0000-0001-9141-7773 - Related articles
-
Severity of microvascular invasion does matter in hepatocellular carcinoma prognosis



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement1
Supplement1 Print
Print



