| Clin Mol Hepatol > Volume 28(3); 2022 > Article |
|
ABSTRACT
Background/Aims
Methods
Results
Conclusions
ACKNOWLEDGMENTS
FOOTNOTES
SUPPLEMENTAL MATERIAL
Supplementary Table 1.
Supplementary Table 2.
Supplementary Table 3.
Supplementary Table 4.
Supplementary Table 5.
Supplementary Table 6.
Supplementary Figure 1.
Figure 1.
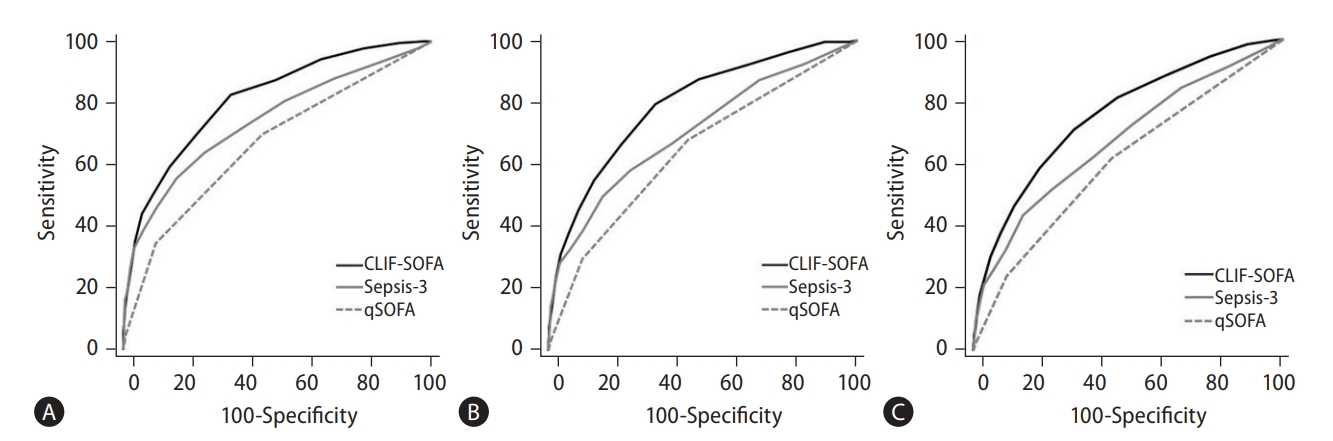
Figure 2.
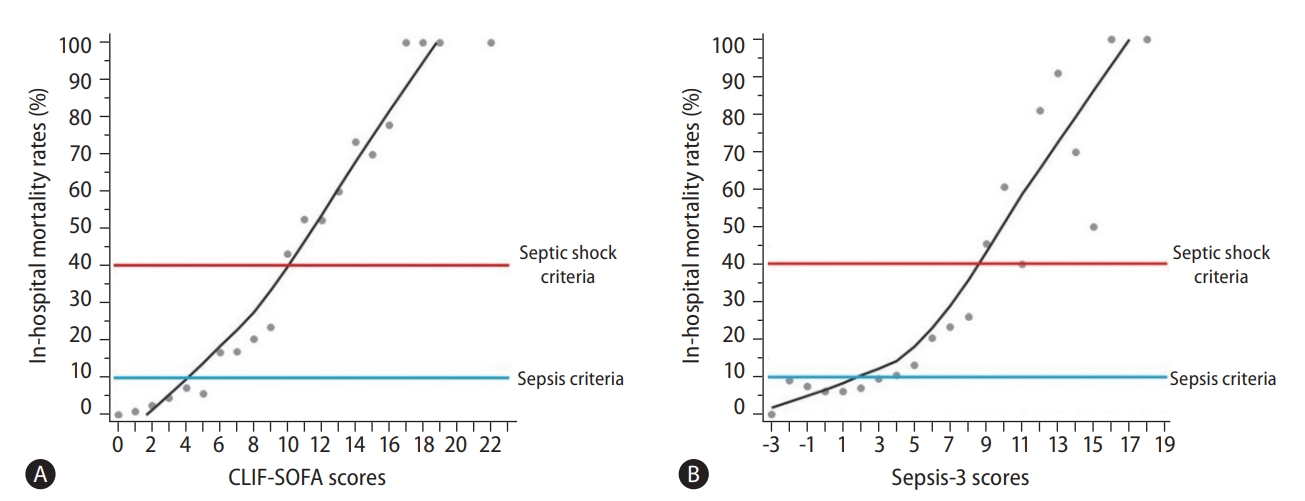
Figure 3.

Table 1.
| Variable | Total population (n=1,622) | In-hospital mortality (-) (n=1,378) | In-hospital mortality (+) (n=244) | P-value‡ |
|---|---|---|---|---|
| Age (years) | 60±13 | 60±13 | 62±14 | 0.04 |
| Gender, male | 1,202 (74.1) | 1,012 (73.4) | 190 (77.9) | 0.15 |
| Etiology of cirrhosis | 0.40 | |||
| Alcohol | 949 (58.5) | 809 (58.7) | 140 (57.4) | |
| HBV | 338 (20.8) | 294 (18.1) | 44 (18.0) | |
| HCV | 91 (5.6) | 74 (5.4) | 17 (7.0) | |
| NAFLD | 218 (13.4) | 181 (13.1) | 37 (15.2) | |
| Others* | 26 (1.6) | 20 (1.5) | 6 (2.5) | |
| Diabetes | 491 (30.3) | 429 (31.1) | 62 (25.4) | 0.08 |
| Body mass index (kg/m2) | 22.8 (20.6–25.4) | 22.9 (20.6–25.4) | 22.3 (20.4–25.0) | 0.21 |
| Mean arterial pressure (mmHg) | 87 (74–101) | 89 (76–102) | 79 (65–94) | <0.001 |
| Heart rate (beat/minute) | 95 (81–111) | 94 (80–110) | 100 (84–116) | 0.002 |
| Body temperature (℃) | 36.8 (36.2–37.5) | 36.8 (36.3–37.5) | 36.5 (35.9–37.2) | <0.001 |
| Respiratory rate (breath/min) | 20 (18–20) | 20 (18–20) | 20 (19–22) | <0.001 |
| Ascites | 734 (45.3) | 607 (44.0) | 127 (52.0) | 0.02 |
| Hepatic encephalopathy | 330 (20.3) | 239 (17.3) | 91 (37.3) | <0.001 |
| ACLF | 601 (37.1) | 411 (29.8) | 190 (77.9) | <0.001 |
| ACLF grade† | <0.001 | |||
| No ACLF | 1,021 (62.9) | 967 (70.2) | 54 (22.1) | |
| Grade 1 | 401 (24.7) | 314 (22.8) | 87 (35.7) | |
| Grade 2 | 132 (8.1) | 76 (5.5) | 56 (23.0) | |
| Grade 3 | 68 (4.2) | 21 (1.5) | 47 (19.3) | |
| INR | 1.4 (1.2–1.6) | 1.3 (1.2–1.6) | 1.7 (1.4–2.3) | <0.001 |
| Total bilirubin (mg/dL) | 2.1 (1.1–4.3) | 1.9 (1.0–3.8) | 3.8 (1.8–8.6) | <0.001 |
| Albumin (g/dL) | 2.9 (2.5–3.4) | 3.0 (2.6–3.5) | 2.4 (2.0–2.9) | <0.001 |
| Creatinine (mg/dL) | 1.0 (0.8–1.6) | 1.0 (0.7–1.4) | 1.8 (1.0–2.9) | <0.001 |
| Sodium (mEq/L) | 136 (131–139) | 136 (132–139) | 134 (128–138) | <0.001 |
| Platelet (×109/L) | 103 (69–159) | 104 (69–158) | 92 (60–160) | 0.04 |
| Leukocyte (×109/L) | 8.0 (5.5–12.0) | 7.8 (5.4–11.3) | 10.0 (6.0–14.7) | <0.001 |
| C-reactive protein (mg/L) | 23 (8–73) | 20 (8–68) | 47 (13–106) | <0.001 |
| MELD score | 15 (11–21) | 14 (11–19) | 23 (18–31) | <0.001 |
| Child-Pugh score | 8 (6–9) | 7 (6–9) | 9 (8–11) | <0.001 |
| SOFA score before ER visit§ | 2 (1–4) | 1 (1–4) | 2 (1–4) | 0.006 |
| SOFA score | 4 (3–6) | 4 (2–6) | 7 (5–11) | <0.001 |
| Sepsis-3 criteria (increase in SOFA ≥2) | 1,160 (71.5) | 945 (68.6) | 215 (88.1) | <0.001 |
| qSOFA ≥2 | 231 (14.2) | 146 (10.6) | 85 (34.8) | <0.001 |
| CLIF-SOFA score† | 5 (3–7) | 4 (3–6) | 9 (6–12) | <0.001 |
| CLIF-C-AD score† | 56.1 (50.0–63.6) | 54.9 (49.3–61.6) | 64.9 (57.1–73.1) | <0.001 |
Values are presented as mean±standard deviation, median (interquartile range), or number (%).
HBV, hepatitis B virus; HCV, hepatitis C virus; NAFLD, nonalcoholic fatty liver disease; ACLF, acute-on-chronic liver failure; INR, international normalized ratio for prothrombin time; MELD, model for end-stage liver disease; SOFA, Sequential Organ Failure Assessment; ER, emergency room; qSOFA, quick Sequential Organ Failure Assessment; CLIF-SOFA, Chronic Liver Failure-Sequential Organ Failure Assessment; CLIF-C-AD, Chronic Liver Failure Consortium Acute Decompensation score.
* Others included the causes of autoimmune hepatitis, primary biliary cholangitis, and cryptogenic cirrhosis.
† ACLF defined according to the European Association for the Study of the Liver-Chronic Liver Failure Consortium (EASL-CLIF).
Table 2.
| Variable | Value (n=1,622) |
|---|---|
| Site of infection | |
| Abdominal | 994 (61.3) |
| Pulmonary | 247 (15.2) |
| Urinary tract | 192 (11.8) |
| Skin and soft tissue | 70 (4.3) |
| Catheter-related | 5 (0.3) |
| Others | 114 (7.0) |
| Type of infection | |
| Community-acquired | 1,466 (90.4) |
| Healthcare-associated | 156 (9.6) |
| Positive qSOFA score | 231 (14.2) |
| Sepsis according to Sepsis-3 criteria | 1,160 (71.5) |
| Septic shock | 87 (5.4) |
| Sepsis* according to in-hospital mortality of 10% | 663 (40.9) |
| MELD score | 19 (13–26) |
| Positive microbiological cultures | 399 (24.6) |
| Type of strains isolated | |
| Gram-positive | 109 (27.3) |
| Gram-negative | 288 (72.2) |
| Fungi | 2 (0.5) |
| Type of bacteria isolated | |
| Escherichia coli | 170 (42.6) |
| Klebsiella pneumoniae | 66 (16.5) |
| Enterococcus faecium | 3 (0.8) |
| Enterococcus faecalis | 13 (3.3) |
| Staphylococcus aureus | 49 (12.3) |
| Pseudomonas aeruginosa | 10 (2.5) |
| Other Enterobacteriaceae | 10 (2.5) |
| Other Streptococci | 39 (9.8) |
| Multimicrobial | 16 (4.0) |
| Others | 26 (6.5) |
| Multidrug-resistant bacteria | 99 (24.8) |
Table 3.
| Prediction model | AUROC (95% CI) | P-value* | Sensitivity (%) | Specificity (%) |
|---|---|---|---|---|
| In-hospital mortality | ||||
| CLIF-SOFA (cut-off=6) | 0.80 (0.78–0.82) | Reference | 66.0 | 78.7 |
| Sepsis-3 (cut-off=2) | 0.75 (0.72–0.77) | <0.001 | 64.2 | 74.8 |
| qSOFA (cut-off=2) | 0.67 (0.64–0.70) | <0.001 | 39.6 | 86.7 |
| Static SOFA (cut-off=3) | 0.78 (0.74–0.71) | <0.001 | 69.7 | 73.6 |
| 1-month mortality | ||||
| CLIF-SOFA (cut-off=6) | 0.77 (0.75–0.80) | Reference | 61.3 | 78.8 |
| Sepsis-3 (cut-off=2) | 0.69 (0.66–0.71) | <0.001 | 57.1 | 74.1 |
| qSOFA (cut-off=2) | 0.63 (0.61–0.66) | <0.001 | 71.3 | 48.2 |
| Static SOFA (cut-off=3) | 0.75 (0.72–0.79) | <0.001 | 65.5 | 73.7 |
| 3-month mortality | ||||
| CLIF-SOFA (cut-off=6) | 0.75 (0.72–0.77) | Reference | 66.1 | 69.9 |
| Sepsis-3 (cut-off=2) | 0.66 (0.63–0.69) | <0.001 | 51.6 | 75.8 |
| qSOFA (cut-off=2) | 0.60 (0.57–0.63) | <0.001 | 66.4 | 48.6 |
| Static SOFA (cut-off=3) | 0.71 (0.68–0.74) | <0.001 | 57.6 | 75.4 |
Abbreviations
REFERENCES
- TOOLS
-
METRICS

- ORCID iDs
-
Minjong Lee

https://orcid.org/0000-0002-3159-5444Dae Hee Choi

https://orcid.org/0000-0002-8956-7518 - Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Supplement1
Supplement1 Print
Print



